| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cathepsin D |
|---|
| Ligand | BDBM335454 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Cathepsin-D Assay |
|---|
| Ki | 3378±n/a nM |
|---|
| Citation |  Walsh, SP; Cumming, JN; He, S; Taoka, BM; Truong, QT; Wu, W C2-carbocyclic iminothiazine dioxides as BACE inhibitors, compositions, and their use US Patent US9732088 Publication Date 8/15/2017 Walsh, SP; Cumming, JN; He, S; Taoka, BM; Truong, QT; Wu, W C2-carbocyclic iminothiazine dioxides as BACE inhibitors, compositions, and their use US Patent US9732088 Publication Date 8/15/2017 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cathepsin D |
|---|
| Name: | Cathepsin D |
|---|
| Synonyms: | CATD_HUMAN | CPSD | CTSD | Cathepsin D [Precursor] | Cathepsin D heavy chain | Cathepsin D light chain | Cathepsin D precursor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 44551.72 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human proCathepsin D (SwissProt accession number P07339) was expressed in Sf9 cells, purified, and autoactivated. |
|---|
| Residue: | 412 |
|---|
| Sequence: | MQPSSLLPLALCLLAAPASALVRIPLHKFTSIRRTMSEVGGSVEDLIAKGPVSKYSQAVP
AVTEGPIPEVLKNYMDAQYYGEIGIGTPPQCFTVVFDTGSSNLWVPSIHCKLLDIACWIH
HKYNSDKSSTYVKNGTSFDIHYGSGSLSGYLSQDTVSVPCQSASSASALGGVKVERQVFG
EATKQPGITFIAAKFDGILGMAYPRISVNNVLPVFDNLMQQKLVDQNIFSFYLSRDPDAQ
PGGELMLGGTDSKYYKGSLSYLNVTRKAYWQVHLDQVEVASGLTLCKEGCEAIVDTGTSL
MVGPVDEVRELQKAIGAVPLIQGEYMIPCEKVSTLPAITLKLGGKGYKLSPEDYTLKVSQ
AGKTLCLSGFMGMDIPPPSGPLWILGDVFIGRYYTVFDRDNNRVGFAEAARL
|
|
|
|---|
| BDBM335454 |
|---|
| n/a |
|---|
| Name | BDBM335454 |
|---|
| Synonyms: | (3R,6S)-5-amino-3-(5-((2-(but- 2-yn-1-yloxy)pyrido[3,4- b]pyrazin-5-yl)amino)-2- fluoropyridin-3-yl)-6- cyclopropyl-6-(fluoromethyl)- 3-methyl-3,6-dihydro-2H-1,4- thiazine 1,1-dioxide | US9732088, Example 13 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H25F2N7O3S |
|---|
| Mol. Mass. | 541.573 |
|---|
| SMILES | CC#CCOc1cnc2c(Nc3cnc(F)c(c3)[C@]3(C)CS(=O)(=O)[C@@](CF)(C4CC4)C(N)=N3)nccc2n1 |r,c:33| |
|---|
| Structure | 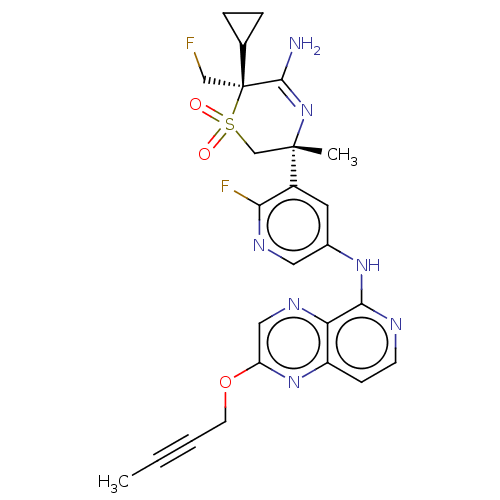
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Walsh, SP; Cumming, JN; He, S; Taoka, BM; Truong, QT; Wu, W C2-carbocyclic iminothiazine dioxides as BACE inhibitors, compositions, and their use US Patent US9732088 Publication Date 8/15/2017
Walsh, SP; Cumming, JN; He, S; Taoka, BM; Truong, QT; Wu, W C2-carbocyclic iminothiazine dioxides as BACE inhibitors, compositions, and their use US Patent US9732088 Publication Date 8/15/2017