| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Nitric oxide synthase, endothelial [N368D] |
|---|
| Ligand | BDBM22030 |
|---|
| Substrate/Competitor | BDBM21959 |
|---|
| Meas. Tech. | NOS Enzyme Inhibition Assay |
|---|
| pH | 7.5±n/a |
|---|
| Temperature | 303.15±n/a K |
|---|
| Ki | 9500±n/a nM |
|---|
| Citation |  Flinspach, ML; Li, H; Jamal, J; Yang, W; Huang, H; Hah, JM; Giómez-Vidal, JA; Litzinger, EA; Silverman, RB; Poulos, TL Structural basis for dipeptide amide isoform-selective inhibition of neuronal nitric oxide synthase. Nat Struct Mol Biol11:54-9 (2004) [PubMed] Article Flinspach, ML; Li, H; Jamal, J; Yang, W; Huang, H; Hah, JM; Giómez-Vidal, JA; Litzinger, EA; Silverman, RB; Poulos, TL Structural basis for dipeptide amide isoform-selective inhibition of neuronal nitric oxide synthase. Nat Struct Mol Biol11:54-9 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Nitric oxide synthase, endothelial [N368D] |
|---|
| Name: | Nitric oxide synthase, endothelial [N368D] |
|---|
| Synonyms: | Constitutive NOS | Endothelial NOS | Endothelial nitric oxide synthase | NOS type III | NOS3 | NOS3_BOVIN | Nitric Oxide Synthase, endothelial Mutant (N368D) | cNOS |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 133292.24 |
|---|
| Organism: | Bos taurus (bovine) |
|---|
| Description: | Recombinant enzyme overexpressed in E. coli. |
|---|
| Residue: | 1205 |
|---|
| Sequence: | MGNLKSVGQEPGPPCGLGLGLGLGLCGKQGPASPAPEPSRAPAPATPHAPDHSPAPNSPT
LTRPPEGPKFPRVKNWELGSITYDTLCAQSQQDGPCTPRCCLGSLVLPRKLQTRPSPGPP
PAEQLLSQARDFINQYYSSIKRSGSQAHEERLQEVEAEVASTGTYHLRESELVFGAKQAW
RNAPRCVGRIQWGKLQVFDARDCSSAQEMFTYICNHIKYATNRGNLRSAITVFPQRAPGR
GDFRIWNSQLVRYAGYRQQDGSVRGDPANVEITELCIQHGWTPGNGRFDVLPLLLQAPDE
APELFVLPPELVLEVPLEHPTLEWFAALGLRWYALPAVSNMLLEIGGLEFSAAPFSGWYM
STEIGTRDLCDPHRYNILEDVAVCMDLDTRTTSSLWKDKAAVEINLAVLHSFQLAKVTIV
DHHAATVSFMKHLDNEQKARGGCPADWAWIVPPISGSLTPVFHQEMVNYILSPAFRYQPD
PWKGSATKGAGITRKKTFKEVANAVKISASLMGTLMAKRVKATILYASETGRAQSYAQQL
GRLFRKAFDPRVLCMDEYDVVSLEHEALVLVVTSTFGNGDPPENGESFAAALMEMSGPYN
SSPRPEQHKSYKIRFNSVSCSDPLVSSWRRKRKESSNTDSAGALGTLRFCVFGLGSRAYP
HFCAFARAVDTRLEELGGERLLQLGQGDELCGQEEAFRGWAKAAFQASCETFCVGEEAKA
AAQDIFSPKRSWKRQRYRLSTQAEGLQLLPGLIHVHRRKMFQATVLSVENLQSSKSTRAT
ILVRLDTAGQEGLQYQPGDHIGICPPNRPGLVEALLSRVEDPPPPTESVAVEQLEKGSPG
GPPPSWVRDPRLPPCTLRQALTFFLDITSPPSPRLLRLLSTLAEEPSEQQELETLSQDPR
RYEEWKWFRCPTLLEVLEQFPSVALPAPLLLTQLPLLQPRYYSVSSAPNAHPGEVHLTVA
VLAYRTQDGLGPLHYGVCSTWLSQLKTGDPVPCFIRGAPSFRLPPDPYVPCILVGPGTGI
APFRGFWQERLHDIESKGLQPAPMTLVFGCRCSQLDHLYRDEVQDAQERGVFGRVLTAFS
REPDSPKTYVQDILRTELAAEVHRVLCLERGHMFVCGDVTMATSVLQTVQRILATEGDME
LDEAGDVIGVLRDQQRYHEDIFGLTLRTQEVTSRIRTQSFSLQERHLRGAVPWAFDPPGP
DTPGP
|
|
|
|---|
| BDBM22030 |
|---|
| BDBM21959 |
|---|
| Name | BDBM22030 |
|---|
| Synonyms: | (2S)-2-amino-N-[(1S)-3-amino-1-(aminocarbonyl)propyl]-5-{[(E)-imino(nitroamino)methyl]amino}pentanamide | (2S)-2-amino-N-[(1S)-3-amino-1-(aminocarbonyl)propyl]-5-{[(Z)-imino(nitroamino)methyl]amino}pentanamide | (2S)-2-amino-N-[(1S)-3-amino-1-(aminocarbonyl)propyl]-5-{[imino(nitroamino)methyl]amino}pentanamide | (2S)-2-amino-N-[(1S)-3-amino-1-carbamoylpropyl]-5-(1-nitrocarbamimidamido)pentanamide | CHEMBL1204421 | CHEMBL44833 | CHEMBL481413 | JMC522060 Compound 1 | L-Arg NO2-L-Dbu-NH2 | L-N(OMEGA)-NITROARGININE-2,4-L-DIAMINOBUTYRIC AMIDE | L-Nomega-nitroarginine-2,4-L-diaminobutyramide | dipeptide amide inhibitor I |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C10H22N8O4 |
|---|
| Mol. Mass. | 318.3329 |
|---|
| SMILES | NCC[C@H](NC(=O)[C@@H](N)CCCN=C(N)N[N+]([O-])=O)C(N)=O |r,w:12.11| |
|---|
| Structure | 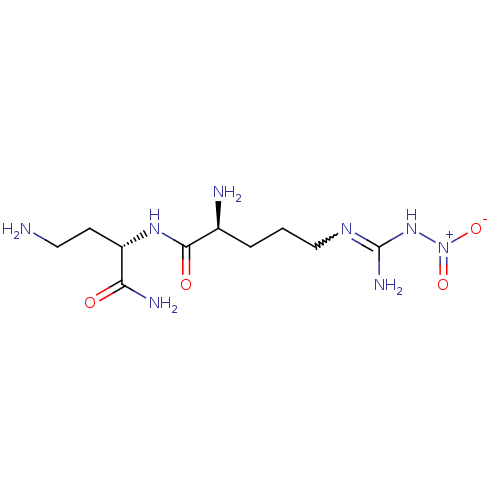
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Flinspach, ML; Li, H; Jamal, J; Yang, W; Huang, H; Hah, JM; Giómez-Vidal, JA; Litzinger, EA; Silverman, RB; Poulos, TL Structural basis for dipeptide amide isoform-selective inhibition of neuronal nitric oxide synthase. Nat Struct Mol Biol11:54-9 (2004) [PubMed] Article
Flinspach, ML; Li, H; Jamal, J; Yang, W; Huang, H; Hah, JM; Giómez-Vidal, JA; Litzinger, EA; Silverman, RB; Poulos, TL Structural basis for dipeptide amide isoform-selective inhibition of neuronal nitric oxide synthase. Nat Struct Mol Biol11:54-9 (2004) [PubMed] Article