

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Histone-lysine N-methyltransferase SMYD3 | ||
| Ligand | BDBM378450 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | SMYD3 Biochemical Assay | ||
| IC50 | 1.10±n/a nM | ||
| Citation |  Foley, MA; Kuntz, KW; Mills, JE; Mitchell, LH; Munchhof, MJ; Harvey, DM Substituted 1,2,3-triazoles as SMYD inhibitors for treating cancer US Patent US10266526 Publication Date 4/23/2019 Foley, MA; Kuntz, KW; Mills, JE; Mitchell, LH; Munchhof, MJ; Harvey, DM Substituted 1,2,3-triazoles as SMYD inhibitors for treating cancer US Patent US10266526 Publication Date 4/23/2019 | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Histone-lysine N-methyltransferase SMYD3 | |||
| Name: | Histone-lysine N-methyltransferase SMYD3 | ||
| Synonyms: | SET and MYND domain-containing protein 3 | SMYD3 | SMYD3_HUMAN | ZMYND1 | ZNFN3A1 | Zinc finger MYND domain-containing protein 1 | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 49101.22 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | Q9H7B4-2 | ||
| Residue: | 428 | ||
| Sequence: |
| ||
| BDBM378450 | |||
| n/a | |||
| Name | BDBM378450 | ||
| Synonyms: | N-((1R,3R,5S)-8-(((1r,4R)-4-aminocyclohexyl)sulfonyl)-8-azabicyclo[3.2.1]octan-3-yl)-6-chloro-2-oxoindoline-5-carboxamide | US10266526, Compound 583 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C22H29ClN4O4S | ||
| Mol. Mass. | 481.008 | ||
| SMILES | N[C@H]1CC[C@@H](CC1)S(=O)(=O)N1[C@H]2CC[C@@H]1C[C@@H](C2)NC(=O)c1cc2CC(=O)Nc2cc1Cl |r,wU:1.0,14.15,11.11,wD:4.7,16.20,TLB:7:10:13.12:17.16.15,(-9.98,-1.26,;-8.44,-1.26,;-7.67,.07,;-6.13,.07,;-5.36,-1.26,;-6.13,-2.6,;-7.67,-2.6,;-3.82,-1.26,;-3.82,-2.8,;-3.82,.28,;-2.28,-1.26,;-1.88,-3,;-3.08,-2.04,;-3.08,-.5,;-1.88,.46,;-.38,.12,;.29,-1.27,;-.38,-2.66,;1.83,-1.27,;2.6,.06,;1.83,1.4,;4.14,.06,;4.91,1.4,;6.45,1.4,;7.48,2.54,;8.89,1.91,;9.98,3,;8.73,.38,;7.22,.06,;6.45,-1.27,;4.91,-1.27,;4.14,-2.61,)| | ||
| Structure | 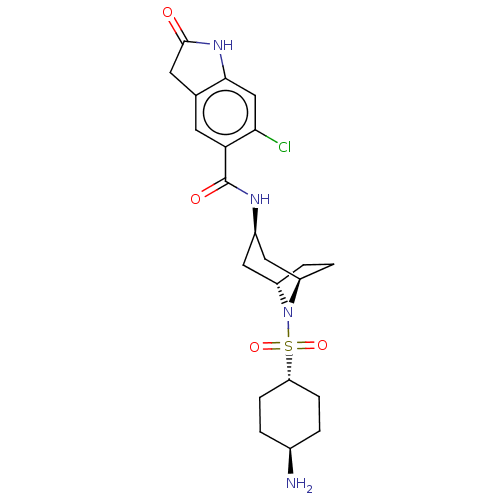
| ||