| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glycogen phosphorylase, liver form |
|---|
| Ligand | BDBM24393 |
|---|
| Substrate/Competitor | BDBM24362 |
|---|
| Meas. Tech. | Enzyme Inhibition Assay |
|---|
| IC50 | 2±n/a nM |
|---|
| Citation |  Birch, AM; Kenny, PW; Oikonomakos, NG; Otterbein, L; Schofield, P; Whittamore, PR; Whalley, DP Development of potent, orally active 1-substituted-3,4-dihydro-2-quinolone glycogen phosphorylase inhibitors. Bioorg Med Chem Lett17:394-9 (2007) [PubMed] Article Birch, AM; Kenny, PW; Oikonomakos, NG; Otterbein, L; Schofield, P; Whittamore, PR; Whalley, DP Development of potent, orally active 1-substituted-3,4-dihydro-2-quinolone glycogen phosphorylase inhibitors. Bioorg Med Chem Lett17:394-9 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Glycogen phosphorylase, liver form |
|---|
| Name: | Glycogen phosphorylase, liver form |
|---|
| Synonyms: | Glycogen Phosphorylase (PYGL) | Glycogen Phosphorylase, liver form | Liver glycogen phosphorylase | PYGL | PYGL_HUMAN |
|---|
| Type: | Homodimer |
|---|
| Mol. Mass.: | 97153.98 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Dimers associate into a tetramer to form the enzymatically active phosphorylase A. |
|---|
| Residue: | 847 |
|---|
| Sequence: | MAKPLTDQEKRRQISIRGIVGVENVAELKKSFNRHLHFTLVKDRNVATTRDYYFALAHTV
RDHLVGRWIRTQQHYYDKCPKRVYYLSLEFYMGRTLQNTMINLGLQNACDEAIYQLGLDI
EELEEIEEDAGLGNGGLGRLAACFLDSMATLGLAAYGYGIRYEYGIFNQKIRDGWQVEEA
DDWLRYGNPWEKSRPEFMLPVHFYGKVEHTNTGTKWIDTQVVLALPYDTPVPGYMNNTVN
TMRLWSARAPNDFNLRDFNVGDYIQAVLDRNLAENISRVLYPNDNFFEGKELRLKQEYFV
VAATLQDIIRRFKASKFGSTRGAGTVFDAFPDQVAIQLNDTHPALAIPELMRIFVDIEKL
PWSKAWELTQKTFAYTNHTVLPEALERWPVDLVEKLLPRHLEIIYEINQKHLDRIVALFP
KDVDRLRRMSLIEEEGSKRINMAHLCIVGSHAVNGVAKIHSDIVKTKVFKDFSELEPDKF
QNKTNGITPRRWLLLCNPGLAELIAEKIGEDYVKDLSQLTKLHSFLGDDVFLRELAKVKQ
ENKLKFSQFLETEYKVKINPSSMFDVQVKRIHEYKRQLLNCLHVITMYNRIKKDPKKLFV
PRTVIIGGKAAPGYHMAKMIIKLITSVADVVNNDPMVGSKLKVIFLENYRVSLAEKVIPA
TDLSEQISTAGTEASGTGNMKFMLNGALTIGTMDGANVEMAEEAGEENLFIFGMRIDDVA
ALDKKGYEAKEYYEALPELKLVIDQIDNGFFSPKQPDLFKDIINMLFYHDRFKVFADYEA
YVKCQDKVSQLYMNPKAWNTMVLKNIAASGKFSSDRTIKEYAQNIWNVEPSDLKISLSNE
SNKVNGN
|
|
|
|---|
| BDBM24393 |
|---|
| BDBM24362 |
|---|
| Name | BDBM24393 |
|---|
| Synonyms: | 2,3-dichloro-N-{1-[(2R)-2,3-dihydroxypropyl]-2-oxo-1,2,3,4-tetrahydroquinolin-3-yl}-4H-thieno[3,2-b]pyrrole-5-carboxamide | 3,4-dihydro-2-quinolone (DHQ) derivative, 32 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H17Cl2N3O4S |
|---|
| Mol. Mass. | 454.327 |
|---|
| SMILES | OC[C@H](O)CN1C(=O)C(Cc2ccccc12)NC(=O)c1cc2sc(Cl)c(Cl)c2[nH]1 |r| |
|---|
| Structure | 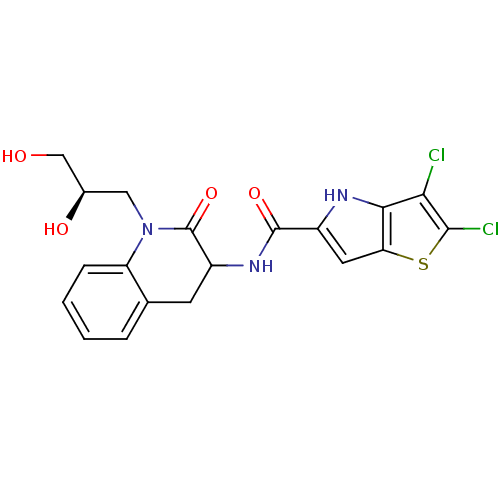
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Birch, AM; Kenny, PW; Oikonomakos, NG; Otterbein, L; Schofield, P; Whittamore, PR; Whalley, DP Development of potent, orally active 1-substituted-3,4-dihydro-2-quinolone glycogen phosphorylase inhibitors. Bioorg Med Chem Lett17:394-9 (2007) [PubMed] Article
Birch, AM; Kenny, PW; Oikonomakos, NG; Otterbein, L; Schofield, P; Whittamore, PR; Whalley, DP Development of potent, orally active 1-substituted-3,4-dihydro-2-quinolone glycogen phosphorylase inhibitors. Bioorg Med Chem Lett17:394-9 (2007) [PubMed] Article