| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2C |
|---|
| Ligand | BDBM35247 |
|---|
| Substrate/Competitor | BDBM28582 |
|---|
| Meas. Tech. | Norepinephrine Uptake Assay (IC50) |
|---|
| Ki | 2400±n/a nM |
|---|
| Citation |  Heffernan, GD; Coghlan, RD; Manas, ES; McDevitt, RE; Li, Y; Mahaney, PE; Robichaud, AJ; Huselton, C; Alfinito, P; Bray, JA; Cosmi, SA; Johnston, GH; Kenney, T; Koury, E; Winneker, RC; Deecher, DC; Trybulski, EJ Dual acting norepinephrine reuptake inhibitors and 5-HT(2A) receptor antagonists: Identification, synthesis and activity of novel 4-aminoethyl-3-(phenylsulfonyl)-1H-indoles. Bioorg Med Chem17:7802-15 (2009) [PubMed] Article Heffernan, GD; Coghlan, RD; Manas, ES; McDevitt, RE; Li, Y; Mahaney, PE; Robichaud, AJ; Huselton, C; Alfinito, P; Bray, JA; Cosmi, SA; Johnston, GH; Kenney, T; Koury, E; Winneker, RC; Deecher, DC; Trybulski, EJ Dual acting norepinephrine reuptake inhibitors and 5-HT(2A) receptor antagonists: Identification, synthesis and activity of novel 4-aminoethyl-3-(phenylsulfonyl)-1H-indoles. Bioorg Med Chem17:7802-15 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2C |
|---|
| Name: | 5-hydroxytryptamine receptor 2C |
|---|
| Synonyms: | 5-HT-1C | 5-HT-2C | 5-HT1C | 5-HT2C | 5-HT2C-INI | 5-HT2c VGI | 5-HTR2C | 5-hydroxytryptamine receptor 1C | 5-hydroxytryptamine receptor 2C (5-HT-2C) | 5-hydroxytryptamine receptor 2C (5HT-2C) | 5HT-1C | 5HT2C_HUMAN | HTR1C | HTR2C | Serotonin (5-HT3) receptor | Serotonin 2c (5-HT2c) receptor | Serotonin Receptor 2C |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 51836.79 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28335 |
|---|
| Residue: | 458 |
|---|
| Sequence: | MVNLRNAVHSFLVHLIGLLVWQSDISVSPVAAIVTDIFNTSDGGRFKFPDGVQNWPALSI
VIIIIMTIGGNILVIMAVSMEKKLHNATNYFLMSLAIADMLVGLLVMPLSLLAILYDYVW
PLPRYLCPVWISLDVLFSTASIMHLCAISLDRYVAIRNPIEHSRFNSRTKAIMKIAIVWA
ISIGVSVPIPVIGLRDEEKVFVNNTTCVLNDPNFVLIGSFVAFFIPLTIMVITYCLTIYV
LRRQALMLLHGHTEEPPGLSLDFLKCCKRNTAEEENSANPNQDQNARRRKKKERRPRGTM
QAINNERKASKVLGIVFFVFLIMWCPFFITNILSVLCEKSCNQKLMEKLLNVFVWIGYVC
SGINPLVYTLFNKIYRRAFSNYLRCNYKVEKKPPVRQIPRVAATALSGRELNVNIYRHTN
EPVIEKASDNEPGIEMQVENLELPVNPSSVVSERISSV
|
|
|
|---|
| BDBM35247 |
|---|
| BDBM28582 |
|---|
| Name | BDBM35247 |
|---|
| Synonyms: | 3-(phenylsulfonyl)-1H-indole, 18i |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H24N2O2S |
|---|
| Mol. Mass. | 368.492 |
|---|
| SMILES | O=S(=O)(c1c[nH]c2cccc(CCN3CCCCC3)c12)c1ccccc1 |
|---|
| Structure | 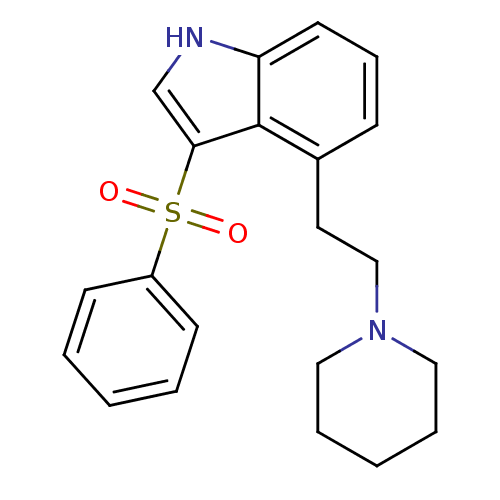
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Heffernan, GD; Coghlan, RD; Manas, ES; McDevitt, RE; Li, Y; Mahaney, PE; Robichaud, AJ; Huselton, C; Alfinito, P; Bray, JA; Cosmi, SA; Johnston, GH; Kenney, T; Koury, E; Winneker, RC; Deecher, DC; Trybulski, EJ Dual acting norepinephrine reuptake inhibitors and 5-HT(2A) receptor antagonists: Identification, synthesis and activity of novel 4-aminoethyl-3-(phenylsulfonyl)-1H-indoles. Bioorg Med Chem17:7802-15 (2009) [PubMed] Article
Heffernan, GD; Coghlan, RD; Manas, ES; McDevitt, RE; Li, Y; Mahaney, PE; Robichaud, AJ; Huselton, C; Alfinito, P; Bray, JA; Cosmi, SA; Johnston, GH; Kenney, T; Koury, E; Winneker, RC; Deecher, DC; Trybulski, EJ Dual acting norepinephrine reuptake inhibitors and 5-HT(2A) receptor antagonists: Identification, synthesis and activity of novel 4-aminoethyl-3-(phenylsulfonyl)-1H-indoles. Bioorg Med Chem17:7802-15 (2009) [PubMed] Article