| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Anoctamin-2 |
|---|
| Ligand | BDBM50239459 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1666073 (CHEMBL4015869) |
|---|
| IC50 | 400±n/a nM |
|---|
| Citation |  Truong, EC; Phuan, PW; Reggi, AL; Ferrera, L; Galietta, LJV; Levy, SE; Moises, AC; Cil, O; Diez-Cecilia, E; Lee, S; Verkman, AS; Anderson, MO Substituted 2-Acylaminocycloalkylthiophene-3-carboxylic Acid Arylamides as Inhibitors of the Calcium-Activated Chloride Channel Transmembrane Protein 16A (TMEM16A). J Med Chem60:4626-4635 (2017) [PubMed] Article Truong, EC; Phuan, PW; Reggi, AL; Ferrera, L; Galietta, LJV; Levy, SE; Moises, AC; Cil, O; Diez-Cecilia, E; Lee, S; Verkman, AS; Anderson, MO Substituted 2-Acylaminocycloalkylthiophene-3-carboxylic Acid Arylamides as Inhibitors of the Calcium-Activated Chloride Channel Transmembrane Protein 16A (TMEM16A). J Med Chem60:4626-4635 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Anoctamin-2 |
|---|
| Name: | Anoctamin-2 |
|---|
| Synonyms: | ANO2 | ANO2_HUMAN | Anoctamin-2 | C12orf3 | TMEM16B | Transmembrane protein 16B |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 113968.03 |
|---|
| Organism: | Homo sapiens |
|---|
| Description: | ChEMBL_117372 |
|---|
| Residue: | 1003 |
|---|
| Sequence: | MATPGPRDIPLLPGSPRRLSPQAGSRGGQGPKHGQQCLKMPGPRAPGLQGGSNRDPGQPC
GGESTRSSSVINNYLDANEPVSLEARLSRMHFHDSQRKVDYVLAYHYRKRGVHLAQGFPG
HSLAIVSNGETGKEPHAGGPGDIELGPLDALEEERKEQREEFEHNLMEAGLELEKDLENK
SQGSIFVRIHAPWQVLAREAEFLKIKVPTKKEMYEIKAGGSIAKKFSAALQKLSSHLQPR
VPEHSNNKMKNLSYPFSREKMYLYNIQEKDTFFDNATRSRIVHEILKRTACSRANNTMGI
NSLIANNIYEAAYPLHDGEYDSPEDDMNDRKLLYQEWARYGVFYKFQPIDLIRKYFGEKI
GLYFAWLGLYTSFLIPSSVIGVIVFLYGCATIEEDIPSREMCDQQNAFTMCPLCDKSCDY
WNLSSACGTAQASHLFDNPATVFFSIFMALWATMFLENWKRLQMRLGYFWDLTGIEEEEE
RAQEHSRPEYETKVREKMLKESNQSAVQKLETNTTECGDEDDEDKLTWKDRFPGYLMNFA
SILFMIALTFSIVFGVIVYRITTAAALSLNKATRSNVRVTVTATAVIINLVVILILDEIY
GAVAKWLTKIEVPKTEQTFEERLILKAFLLKFVNAYSPIFYVAFFKGRFVGRPGSYVYVF
DGYRMEECAPGGCLMELCIQLSIIMLGKQLIQNNIFEIGVPKLKKLFRKLKDETEAGETD
SAHSKHPEQWDLDYSLEPYTGLTPEYMEMIIQFGFVTLFVASFPLAPVFALLNNVIEVRL
DAKKFVTELRRPDAVRTKDIGIWFDILSGIGKFSVISNAFVIAITSDFIPRLVYQYSYSH
NGTLHGFVNHTLSFFNVSQLKEGTQPENSQFDQEVQFCRFKDYREPPWAPNPYEFSKQYW
FILSARLAFVIIFQNLVMFLSVLVDWMIPDIPTDISDQIKKEKSLLVDFFLKEEHEKLKL
MDEPALRSPGGGDRSRSRAASSAPSGQSQLGSMMSSGSQHTNV
|
|
|
|---|
| BDBM50239459 |
|---|
| n/a |
|---|
| Name | BDBM50239459 |
|---|
| Synonyms: | CHEMBL4074929 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H19BrF2N2O2S |
|---|
| Mol. Mass. | 457.332 |
|---|
| SMILES | Cc1ccccc1NC(=O)c1c(NC(=O)C(F)(F)Br)sc2CCCCCc12 |
|---|
| Structure | 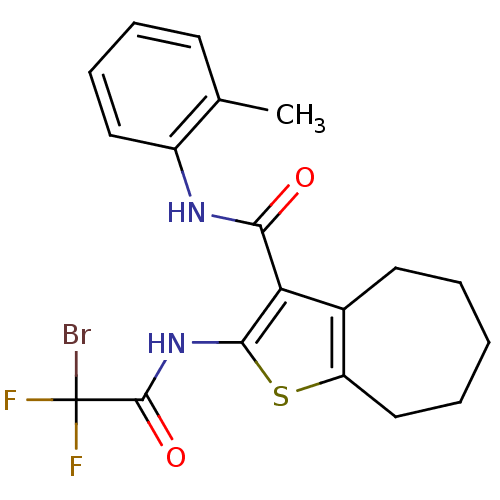
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Truong, EC; Phuan, PW; Reggi, AL; Ferrera, L; Galietta, LJV; Levy, SE; Moises, AC; Cil, O; Diez-Cecilia, E; Lee, S; Verkman, AS; Anderson, MO Substituted 2-Acylaminocycloalkylthiophene-3-carboxylic Acid Arylamides as Inhibitors of the Calcium-Activated Chloride Channel Transmembrane Protein 16A (TMEM16A). J Med Chem60:4626-4635 (2017) [PubMed] Article
Truong, EC; Phuan, PW; Reggi, AL; Ferrera, L; Galietta, LJV; Levy, SE; Moises, AC; Cil, O; Diez-Cecilia, E; Lee, S; Verkman, AS; Anderson, MO Substituted 2-Acylaminocycloalkylthiophene-3-carboxylic Acid Arylamides as Inhibitors of the Calcium-Activated Chloride Channel Transmembrane Protein 16A (TMEM16A). J Med Chem60:4626-4635 (2017) [PubMed] Article