| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Fibroblast growth factor receptor 3 |
|---|
| Ligand | BDBM50240805 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1668739 (CHEMBL4018627) |
|---|
| IC50 | 8.0±n/a nM |
|---|
| Citation |  Zhu, W; Chen, H; Wang, Y; Wang, J; Peng, X; Chen, X; Gao, Y; Li, C; He, Y; Ai, J; Geng, M; Zheng, M; Liu, H Design, Synthesis, and Pharmacological Evaluation of Novel Multisubstituted Pyridin-3-amine Derivatives as Multitargeted Protein Kinase Inhibitors for the Treatment of Non-Small Cell Lung Cancer. J Med Chem60:6018-6035 (2017) [PubMed] Article Zhu, W; Chen, H; Wang, Y; Wang, J; Peng, X; Chen, X; Gao, Y; Li, C; He, Y; Ai, J; Geng, M; Zheng, M; Liu, H Design, Synthesis, and Pharmacological Evaluation of Novel Multisubstituted Pyridin-3-amine Derivatives as Multitargeted Protein Kinase Inhibitors for the Treatment of Non-Small Cell Lung Cancer. J Med Chem60:6018-6035 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Fibroblast growth factor receptor 3 |
|---|
| Name: | Fibroblast growth factor receptor 3 |
|---|
| Synonyms: | CD_antigen: CD333 | FGFR-3 | FGFR3 | FGFR3_HUMAN | Fibroblast growth factor receptor | Fibroblast growth factor receptor 3 (FGFR3) | JTK4 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 87699.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P22607 |
|---|
| Residue: | 806 |
|---|
| Sequence: | MGAPACALALCVAVAIVAGASSESLGTEQRVVGRAAEVPGPEPGQQEQLVFGSGDAVELS
CPPPGGGPMGPTVWVKDGTGLVPSERVLVGPQRLQVLNASHEDSGAYSCRQRLTQRVLCH
FSVRVTDAPSSGDDEDGEDEAEDTGVDTGAPYWTRPERMDKKLLAVPAANTVRFRCPAAG
NPTPSISWLKNGREFRGEHRIGGIKLRHQQWSLVMESVVPSDRGNYTCVVENKFGSIRQT
YTLDVLERSPHRPILQAGLPANQTAVLGSDVEFHCKVYSDAQPHIQWLKHVEVNGSKVGP
DGTPYVTVLKTAGANTTDKELEVLSLHNVTFEDAGEYTCLAGNSIGFSHHSAWLVVLPAE
EELVEADEAGSVYAGILSYGVGFFLFILVVAAVTLCRLRSPPKKGLGSPTVHKISRFPLK
RQVSLESNASMSSNTPLVRIARLSSGEGPTLANVSELELPADPKWELSRARLTLGKPLGE
GCFGQVVMAEAIGIDKDRAAKPVTVAVKMLKDDATDKDLSDLVSEMEMMKMIGKHKNIIN
LLGACTQGGPLYVLVEYAAKGNLREFLRARRPPGLDYSFDTCKPPEEQLTFKDLVSCAYQ
VARGMEYLASQKCIHRDLAARNVLVTEDNVMKIADFGLARDVHNLDYYKKTTNGRLPVKW
MAPEALFDRVYTHQSDVWSFGVLLWEIFTLGGSPYPGIPVEELFKLLKEGHRMDKPANCT
HDLYMIMRECWHAAPSQRPTFKQLVEDLDRVLTVTSTDEYLDLSAPFEQYSPGGQDTPSS
SSSGDDSVFAHDLLPPAPPSSGGSRT
|
|
|
|---|
| BDBM50240805 |
|---|
| n/a |
|---|
| Name | BDBM50240805 |
|---|
| Synonyms: | CHEMBL4073545 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H24N4O2 |
|---|
| Mol. Mass. | 436.5051 |
|---|
| SMILES | Cc1n[nH]c2ccc(cc12)-c1cc(N[C@@H](CO)c2ccccc2)cnc1-c1ccccc1O |r| |
|---|
| Structure | 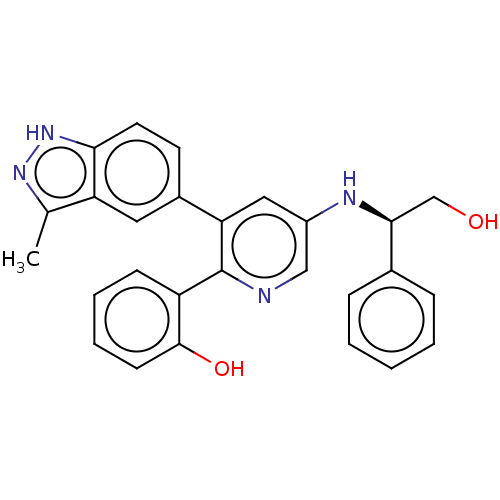
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhu, W; Chen, H; Wang, Y; Wang, J; Peng, X; Chen, X; Gao, Y; Li, C; He, Y; Ai, J; Geng, M; Zheng, M; Liu, H Design, Synthesis, and Pharmacological Evaluation of Novel Multisubstituted Pyridin-3-amine Derivatives as Multitargeted Protein Kinase Inhibitors for the Treatment of Non-Small Cell Lung Cancer. J Med Chem60:6018-6035 (2017) [PubMed] Article
Zhu, W; Chen, H; Wang, Y; Wang, J; Peng, X; Chen, X; Gao, Y; Li, C; He, Y; Ai, J; Geng, M; Zheng, M; Liu, H Design, Synthesis, and Pharmacological Evaluation of Novel Multisubstituted Pyridin-3-amine Derivatives as Multitargeted Protein Kinase Inhibitors for the Treatment of Non-Small Cell Lung Cancer. J Med Chem60:6018-6035 (2017) [PubMed] Article