 Found 6061 hits with Last Name = 'peng' and Initial = 'x'
Found 6061 hits with Last Name = 'peng' and Initial = 'x' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50366764

(CHEMBL1790045 | MCL-117)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC#C)c2c1 Show InChI InChI=1S/C19H23NO/c1-2-10-20-11-9-19-8-4-3-5-16(19)18(20)12-14-6-7-15(21)13-17(14)19/h1,6-7,13,16,18,21H,3-5,8-12H2/t16-,18+,19+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50366764

(CHEMBL1790045 | MCL-117)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC#C)c2c1 Show InChI InChI=1S/C19H23NO/c1-2-10-20-11-9-19-8-4-3-5-16(19)18(20)12-14-6-7-15(21)13-17(14)19/h1,6-7,13,16,18,21H,3-5,8-12H2/t16-,18+,19+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50180190
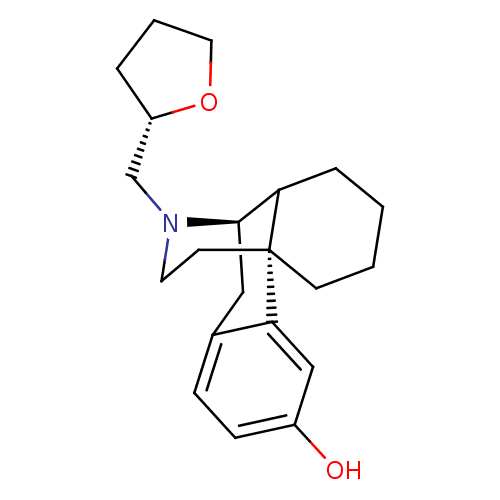
((1R,9R)-17-[(2S)-oxolan-2-ylmethyl]-17-azatetracyc...)Show SMILES Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3C[C@@H]3CCCO3)c2c1 |TLB:16:15:5.22.4:7| Show InChI InChI=1S/C21H29NO2/c23-16-7-6-15-12-20-18-5-1-2-8-21(18,19(15)13-16)9-10-22(20)14-17-4-3-11-24-17/h6-7,13,17-18,20,23H,1-5,8-12,14H2/t17-,18?,20+,21+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50135800
((-)-3-Hydroxy-N-cycloproypylmethylmorphinan Mandel...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CC3)c2c1 Show InChI InChI=1S/C20H27NO/c22-16-7-6-15-11-19-17-3-1-2-8-20(17,18(15)12-16)9-10-21(19)13-14-4-5-14/h6-7,12,14,17,19,22H,1-5,8-11,13H2/t17-,19+,20+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69,593 from human kappa opioid receptor expressed in CHO membrane |
Bioorg Med Chem 15: 4106-12 (2007)
Article DOI: 10.1016/j.bmc.2007.03.076
BindingDB Entry DOI: 10.7270/Q2D50MN1 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50135800

((-)-3-Hydroxy-N-cycloproypylmethylmorphinan Mandel...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CC3)c2c1 Show InChI InChI=1S/C20H27NO/c22-16-7-6-15-11-19-17-3-1-2-8-20(17,18(15)12-16)9-10-21(19)13-14-4-5-14/h6-7,12,14,17,19,22H,1-5,8-11,13H2/t17-,19+,20+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from human kappa opioid receptors expressed in CHO cell membrane |
Bioorg Med Chem Lett 17: 1508-11 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.013
BindingDB Entry DOI: 10.7270/Q2WS8V2W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50105483
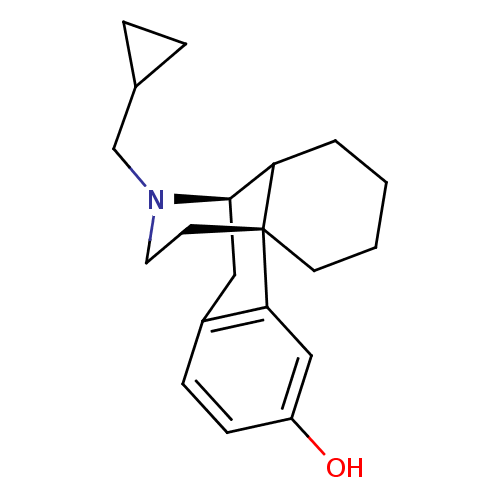
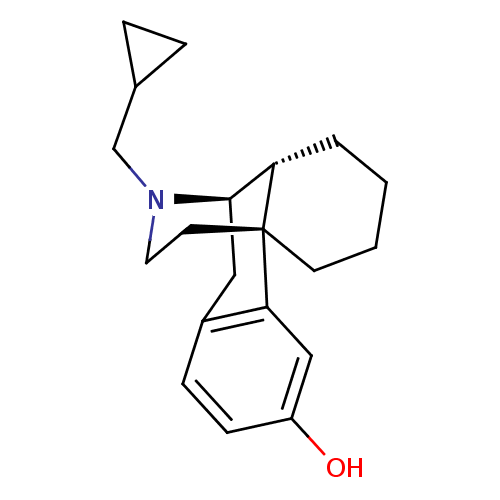
((-)-cyclorphan | 17-cyclopropylmethyl-(1R,9R)-17-a...)Show SMILES Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC3CC3)c2c1 |TLB:8:7:20.4.5:15.13.14| Show InChI InChI=1S/C20H27NO/c22-16-7-6-15-11-19-17-3-1-2-8-20(17,18(15)12-16)9-10-21(19)13-14-4-5-14/h6-7,12,14,17,19,22H,1-5,8-11,13H2/t17?,19-,20-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50204451
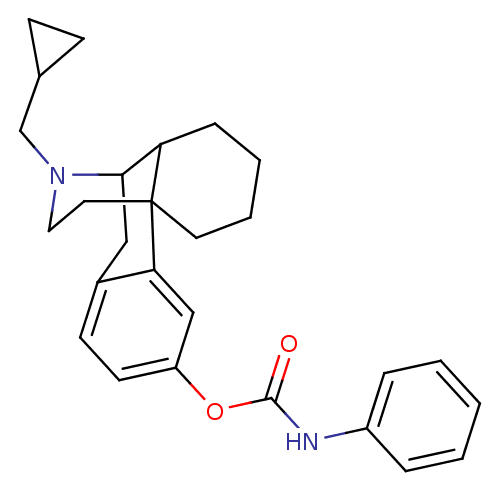
(CHEMBL397035 | MCL-429)Show SMILES O=C(Nc1ccccc1)Oc1ccc2CC3C4CCCCC4(CCN3CC3CC3)c2c1 |TLB:25:24:13.29.14:16,30:29:16:24.22.23| Show InChI InChI=1S/C27H32N2O2/c30-26(28-21-6-2-1-3-7-21)31-22-12-11-20-16-25-23-8-4-5-13-27(23,24(20)17-22)14-15-29(25)18-19-9-10-19/h1-3,6-7,11-12,17,19,23,25H,4-5,8-10,13-16,18H2,(H,28,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from human kappa opioid receptors expressed in CHO cell membrane |
Bioorg Med Chem Lett 17: 1508-11 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.013
BindingDB Entry DOI: 10.7270/Q2WS8V2W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50210557
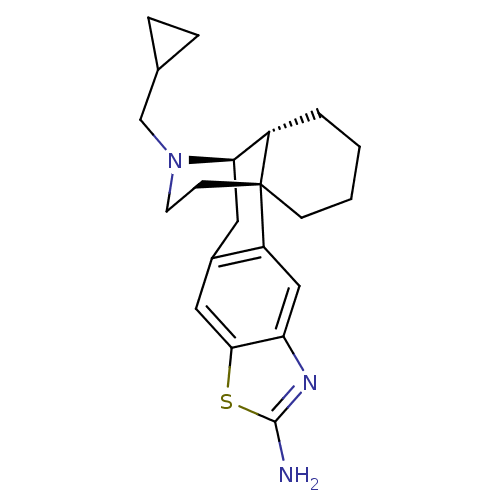
(CHEMBL242756 | MCL-147)Show SMILES Nc1nc2cc3c(C[C@@H]4[C@@H]5CCCC[C@]35CCN4CC3CC3)cc2s1 |THB:18:17:6.5.7:9| Show InChI InChI=1S/C21H27N3S/c22-20-23-17-11-16-14(10-19(17)25-20)9-18-15-3-1-2-6-21(15,16)7-8-24(18)12-13-4-5-13/h10-11,13,15,18H,1-9,12H2,(H2,22,23)/t15-,18+,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69,593 from human kappa opioid receptor expressed in CHO membrane |
Bioorg Med Chem 15: 4106-12 (2007)
Article DOI: 10.1016/j.bmc.2007.03.076
BindingDB Entry DOI: 10.7270/Q2D50MN1 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50135806
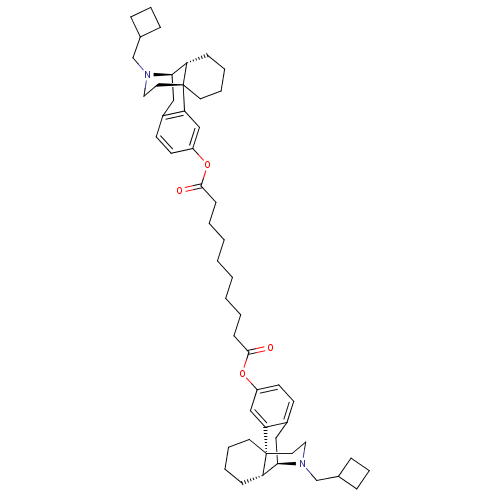
(CHEMBL147511 | MCL-144 | di[17-cyclobutylmethyl-(1...)Show SMILES O=C(CCCCCCCCC(=O)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C52H72N2O4/c55-49(57-41-23-21-39-31-47-43-17-7-9-25-51(43,45(39)33-41)27-29-53(47)35-37-13-11-14-37)19-5-3-1-2-4-6-20-50(56)58-42-24-22-40-32-48-44-18-8-10-26-52(44,46(40)34-42)28-30-54(48)36-38-15-12-16-38/h21-24,33-34,37-38,43-44,47-48H,1-20,25-32,35-36H2/t43-,44-,47+,48+,51+,52+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50204450

(CHEMBL242048 | MCL-428)Show SMILES O=C(Nc1ccccc1)Oc1ccc2CC3C4CCCCC4(CCN3CC3CCC3)c2c1 |TLB:25:24:13.30.14:16,31:30:16:24.22.23| Show InChI InChI=1S/C28H34N2O2/c31-27(29-22-9-2-1-3-10-22)32-23-13-12-21-17-26-24-11-4-5-14-28(24,25(21)18-23)15-16-30(26)19-20-7-6-8-20/h1-3,9-10,12-13,18,20,24,26H,4-8,11,14-17,19H2,(H,29,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from human kappa opioid receptors expressed in CHO cell membrane |
Bioorg Med Chem Lett 17: 1508-11 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.013
BindingDB Entry DOI: 10.7270/Q2WS8V2W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50135800

((-)-3-Hydroxy-N-cycloproypylmethylmorphinan Mandel...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CC3)c2c1 Show InChI InChI=1S/C20H27NO/c22-16-7-6-15-11-19-17-3-1-2-8-20(17,18(15)12-16)9-10-21(19)13-14-4-5-14/h6-7,12,14,17,19,22H,1-5,8-11,13H2/t17-,19+,20+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptors expressed in CHO cell membrane |
Bioorg Med Chem Lett 17: 1508-11 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.013
BindingDB Entry DOI: 10.7270/Q2WS8V2W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50105483

((-)-cyclorphan | 17-cyclopropylmethyl-(1R,9R)-17-a...)Show SMILES Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC3CC3)c2c1 |TLB:8:7:20.4.5:15.13.14| Show InChI InChI=1S/C20H27NO/c22-16-7-6-15-11-19-17-3-1-2-8-20(17,18(15)12-16)9-10-21(19)13-14-4-5-14/h6-7,12,14,17,19,22H,1-5,8-11,13H2/t17?,19-,20-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50135800

((-)-3-Hydroxy-N-cycloproypylmethylmorphinan Mandel...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CC3)c2c1 Show InChI InChI=1S/C20H27NO/c22-16-7-6-15-11-19-17-3-1-2-8-20(17,18(15)12-16)9-10-21(19)13-14-4-5-14/h6-7,12,14,17,19,22H,1-5,8-11,13H2/t17-,19+,20+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO membrane |
Bioorg Med Chem 15: 4106-12 (2007)
Article DOI: 10.1016/j.bmc.2007.03.076
BindingDB Entry DOI: 10.7270/Q2D50MN1 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50180179
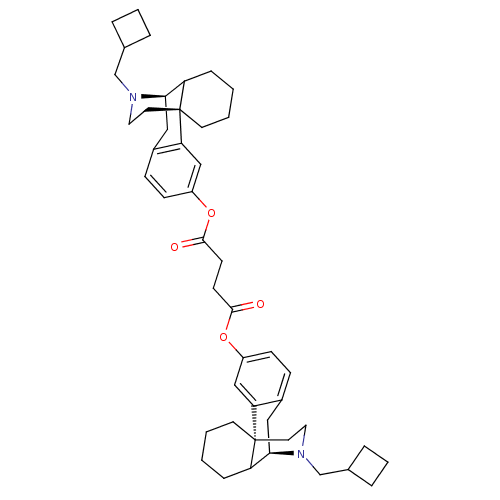
(CHEMBL203251 | MCL-139)Show SMILES O=C(CCC(=O)Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC3CCC3)c2c1)Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C46H60N2O4/c49-43(51-35-15-13-33-25-41-37-11-1-3-19-45(37,39(33)27-35)21-23-47(41)29-31-7-5-8-31)17-18-44(50)52-36-16-14-34-26-42-38-12-2-4-20-46(38,40(34)28-36)22-24-48(42)30-32-9-6-10-32/h13-16,27-28,31-32,37-38,41-42H,1-12,17-26,29-30H2/t37?,38?,41-,42-,45-,46-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0760 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50135808
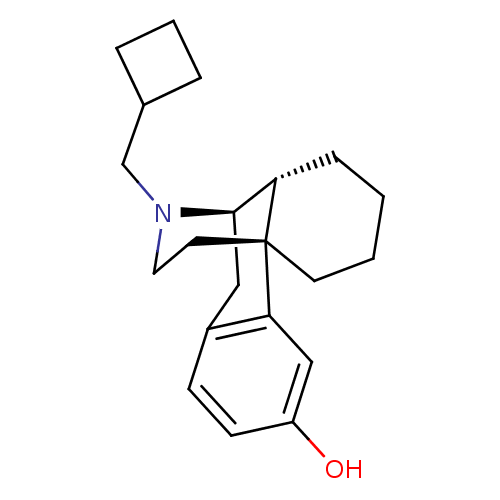
((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 |r,TLB:16:15:4.21.5:7| Show InChI InChI=1S/C21H29NO/c23-17-8-7-16-12-20-18-6-1-2-9-21(18,19(16)13-17)10-11-22(20)14-15-4-3-5-15/h7-8,13,15,18,20,23H,1-6,9-12,14H2/t18-,20+,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0790 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50135808

((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 |r,TLB:16:15:4.21.5:7| Show InChI InChI=1S/C21H29NO/c23-17-8-7-16-12-20-18-6-1-2-9-21(18,19(16)13-17)10-11-22(20)14-15-4-3-5-15/h7-8,13,15,18,20,23H,1-6,9-12,14H2/t18-,20+,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0790 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from human opioid kappa receptor expressed in CHO cells |
J Med Chem 50: 2254-8 (2007)
Article DOI: 10.1021/jm061327z
BindingDB Entry DOI: 10.7270/Q2CZ3804 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50135808

((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 |r,TLB:16:15:4.21.5:7| Show InChI InChI=1S/C21H29NO/c23-17-8-7-16-12-20-18-6-1-2-9-21(18,19(16)13-17)10-11-22(20)14-15-4-3-5-15/h7-8,13,15,18,20,23H,1-6,9-12,14H2/t18-,20+,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0790 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from human kappa opioid receptors expressed in CHO cell membrane |
Bioorg Med Chem Lett 17: 1508-11 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.013
BindingDB Entry DOI: 10.7270/Q2WS8V2W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50135808

((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 |r,TLB:16:15:4.21.5:7| Show InChI InChI=1S/C21H29NO/c23-17-8-7-16-12-20-18-6-1-2-9-21(18,19(16)13-17)10-11-22(20)14-15-4-3-5-15/h7-8,13,15,18,20,23H,1-6,9-12,14H2/t18-,20+,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0790 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69,593 from human kappa opioid receptor expressed in CHO membrane |
Bioorg Med Chem 15: 4106-12 (2007)
Article DOI: 10.1016/j.bmc.2007.03.076
BindingDB Entry DOI: 10.7270/Q2D50MN1 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50135797
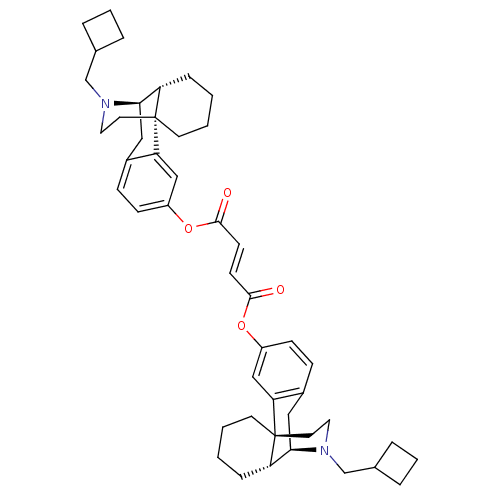
(CHEMBL146756 | di[17-cyclobutylmethyl-(1R,9R,10R)-...)Show SMILES O=C(Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1)\C=C\C(=O)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C46H58N2O4/c49-43(51-35-15-13-33-25-41-37-11-1-3-19-45(37,39(33)27-35)21-23-47(41)29-31-7-5-8-31)17-18-44(50)52-36-16-14-34-26-42-38-12-2-4-20-46(38,40(34)28-36)22-24-48(42)30-32-9-6-10-32/h13-18,27-28,31-32,37-38,41-42H,1-12,19-26,29-30H2/b18-17+/t37-,38-,41+,42+,45+,46+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50135806

(CHEMBL147511 | MCL-144 | di[17-cyclobutylmethyl-(1...)Show SMILES O=C(CCCCCCCCC(=O)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C52H72N2O4/c55-49(57-41-23-21-39-31-47-43-17-7-9-25-51(43,45(39)33-41)27-29-53(47)35-37-13-11-14-37)19-5-3-1-2-4-6-20-50(56)58-42-24-22-40-32-48-44-18-8-10-26-52(44,46(40)34-42)28-30-54(48)36-38-15-12-16-38/h21-24,33-34,37-38,43-44,47-48H,1-20,25-32,35-36H2/t43-,44-,47+,48+,51+,52+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50180192
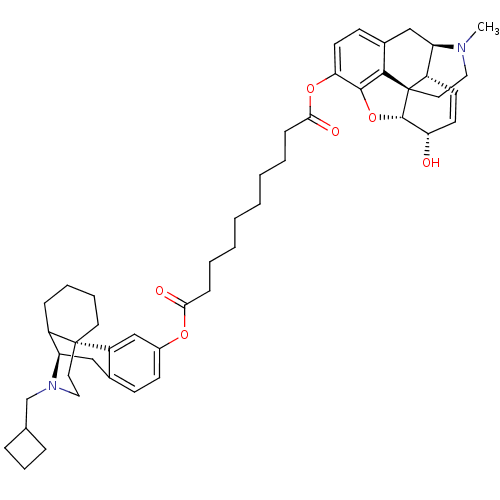
(1-((-)-N-cyclobutylmethylmorphinan-3-yl) 10-(3,6-d...)Show SMILES CN1CC[C@@]23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4O)ccc5OC(=O)CCCCCCCCC(=O)Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC3CCC3)c2c1 |c:16| Show InChI InChI=1S/C48H62N2O6/c1-49-25-24-48-36-19-20-40(51)46(48)56-45-41(21-17-33(44(45)48)28-38(36)49)55-43(53)15-7-5-3-2-4-6-14-42(52)54-34-18-16-32-27-39-35-13-8-9-22-47(35,37(32)29-34)23-26-50(39)30-31-11-10-12-31/h16-21,29,31,35-36,38-40,46,51H,2-15,22-28,30H2,1H3/t35?,36-,38+,39+,40-,46-,47+,48-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50204451

(CHEMBL397035 | MCL-429)Show SMILES O=C(Nc1ccccc1)Oc1ccc2CC3C4CCCCC4(CCN3CC3CC3)c2c1 |TLB:25:24:13.29.14:16,30:29:16:24.22.23| Show InChI InChI=1S/C27H32N2O2/c30-26(28-21-6-2-1-3-7-21)31-22-12-11-20-16-25-23-8-4-5-13-27(23,24(20)17-22)14-15-29(25)18-19-9-10-19/h1-3,6-7,11-12,17,19,23,25H,4-5,8-10,13-16,18H2,(H,28,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptors expressed in CHO cell membrane |
Bioorg Med Chem Lett 17: 1508-11 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.013
BindingDB Entry DOI: 10.7270/Q2WS8V2W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50209670
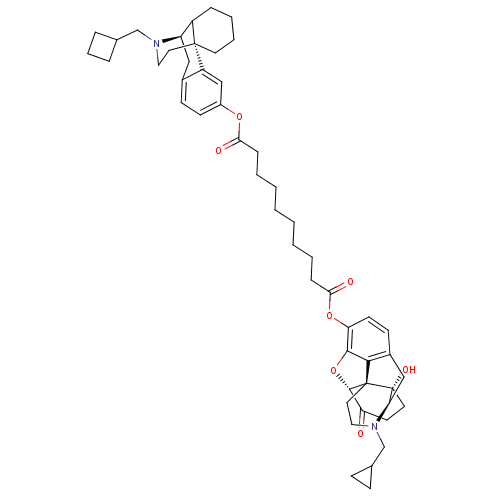
(17-(cyclopropylmethyl)morphinan-3-yl(5alpha)-17-(c...)Show SMILES O[C@]12CCC(=O)[C@@H]3Oc4c5c(C[C@H]1N(CC1CC1)CC[C@@]235)ccc4OC(=O)CCCCCCCCC(=O)Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC3CCC3)c2c1 |TLB:53:52:41.58.42:44| Show InChI InChI=1S/C51H66N2O7/c54-41-21-23-51(57)43-29-36-18-20-42(47-46(36)50(51,48(41)60-47)25-27-53(43)32-34-15-16-34)59-45(56)14-6-4-2-1-3-5-13-44(55)58-37-19-17-35-28-40-38-12-7-8-22-49(38,39(35)30-37)24-26-52(40)31-33-10-9-11-33/h17-20,30,33-34,38,40,43,48,57H,1-16,21-29,31-32H2/t38?,40-,43-,48+,49-,50+,51-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from human opioid kappa receptor expressed in CHO cells |
J Med Chem 50: 2254-8 (2007)
Article DOI: 10.1021/jm061327z
BindingDB Entry DOI: 10.7270/Q2CZ3804 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50204450

(CHEMBL242048 | MCL-428)Show SMILES O=C(Nc1ccccc1)Oc1ccc2CC3C4CCCCC4(CCN3CC3CCC3)c2c1 |TLB:25:24:13.30.14:16,31:30:16:24.22.23| Show InChI InChI=1S/C28H34N2O2/c31-27(29-22-9-2-1-3-10-22)32-23-13-12-21-17-26-24-11-4-5-14-28(24,25(21)18-23)15-16-30(26)19-20-7-6-8-20/h1-3,9-10,12-13,18,20,24,26H,4-8,11,14-17,19H2,(H,29,31) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptors expressed in CHO cell membrane |
Bioorg Med Chem Lett 17: 1508-11 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.013
BindingDB Entry DOI: 10.7270/Q2WS8V2W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50027231
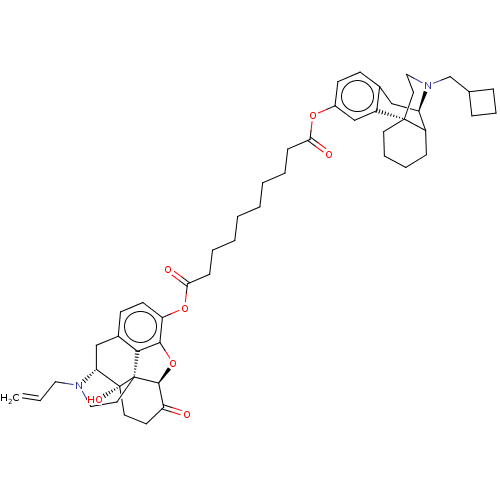
(CHEMBL2113275)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC=C)CC[C@@]14[C@@]5(O)CCC2=O)ccc3OC(=O)CCCCCCCCC(=O)Oc1ccc2C[C@]3([H])C4CCCC[C@]4(CCN3CC3CCC3)c2c1 |TLB:55:54:42.60.43:46,61:60:46:54.52.53,THB:10:9:5.4.6:16| Show InChI InChI=1S/C50H64N2O7/c1-2-26-51-28-25-49-45-35-18-20-41(46(45)59-47(49)40(53)21-23-50(49,56)42(51)30-35)58-44(55)16-8-6-4-3-5-7-15-43(54)57-36-19-17-34-29-39-37-14-9-10-22-48(37,38(34)31-36)24-27-52(39)32-33-12-11-13-33/h2,17-20,31,33,37,39,42,47,56H,1,3-16,21-30,32H2/t37?,39-,42-,47+,48-,49+,50-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from human opioid kappa receptor expressed in CHO cells |
J Med Chem 50: 2254-8 (2007)
Article DOI: 10.1021/jm061327z
BindingDB Entry DOI: 10.7270/Q2CZ3804 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50204449
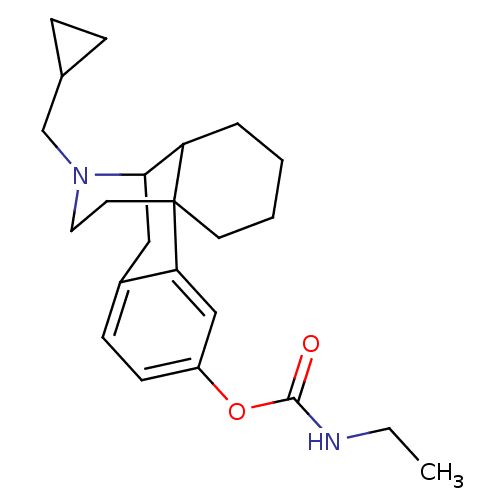
(CHEMBL427862 | MCL-449)Show SMILES CCNC(=O)Oc1ccc2CC3C4CCCCC4(CCN3CC3CC3)c2c1 |TLB:21:20:9.25.10:12,26:25:12:20.18.19| Show InChI InChI=1S/C23H32N2O2/c1-2-24-22(26)27-18-9-8-17-13-21-19-5-3-4-10-23(19,20(17)14-18)11-12-25(21)15-16-6-7-16/h8-9,14,16,19,21H,2-7,10-13,15H2,1H3,(H,24,26) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from human kappa opioid receptors expressed in CHO cell membrane |
Bioorg Med Chem Lett 17: 1508-11 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.013
BindingDB Entry DOI: 10.7270/Q2WS8V2W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50204453
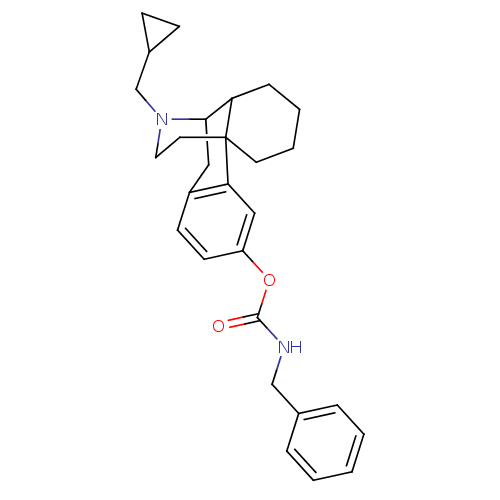
(CHEMBL263096 | MCL-444)Show SMILES O=C(NCc1ccccc1)Oc1ccc2CC3C4CCCCC4(CCN3CC3CC3)c2c1 |TLB:26:25:14.30.15:17,31:30:17:25.23.24| Show InChI InChI=1S/C28H34N2O2/c31-27(29-18-20-6-2-1-3-7-20)32-23-12-11-22-16-26-24-8-4-5-13-28(24,25(22)17-23)14-15-30(26)19-21-9-10-21/h1-3,6-7,11-12,17,21,24,26H,4-5,8-10,13-16,18-19H2,(H,29,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from human kappa opioid receptors expressed in CHO cell membrane |
Bioorg Med Chem Lett 17: 1508-11 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.013
BindingDB Entry DOI: 10.7270/Q2WS8V2W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50180190

((1R,9R)-17-[(2S)-oxolan-2-ylmethyl]-17-azatetracyc...)Show SMILES Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3C[C@@H]3CCCO3)c2c1 |TLB:16:15:5.22.4:7| Show InChI InChI=1S/C21H29NO2/c23-16-7-6-15-12-20-18-5-1-2-8-21(18,19(15)13-16)9-10-22(20)14-17-4-3-11-24-17/h6-7,13,17-18,20,23H,1-5,8-12,14H2/t17-,18?,20+,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50180179

(CHEMBL203251 | MCL-139)Show SMILES O=C(CCC(=O)Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC3CCC3)c2c1)Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C46H60N2O4/c49-43(51-35-15-13-33-25-41-37-11-1-3-19-45(37,39(33)27-35)21-23-47(41)29-31-7-5-8-31)17-18-44(50)52-36-16-14-34-26-42-38-12-2-4-20-46(38,40(34)28-36)22-24-48(42)30-32-9-6-10-32/h13-16,27-28,31-32,37-38,41-42H,1-12,17-26,29-30H2/t37?,38?,41-,42-,45-,46-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Rattus norvegicus (rat)) | BDBM50155461
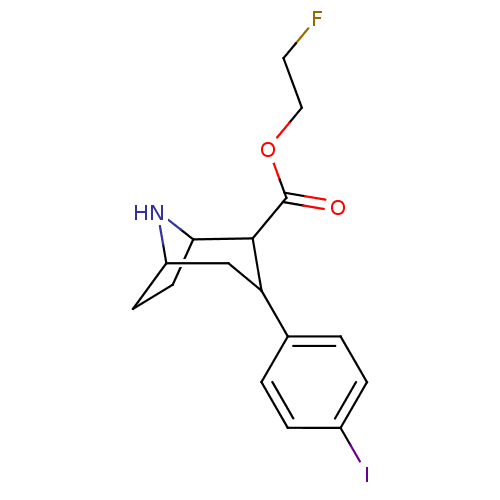
(3-(4-Iodo-phenyl)-8-aza-bicyclo[3.2.1]octane-2-car...)Show SMILES FCCOC(=O)C1C2CCC(CC1c1ccc(I)cc1)N2 |TLB:13:12:20:8.9,THB:4:6:20:8.9| Show InChI InChI=1S/C16H19FINO2/c17-7-8-21-16(20)15-13(9-12-5-6-14(15)19-12)10-1-3-11(18)4-2-10/h1-4,12-15,19H,5-9H2 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]-cyanoimiprimine from serotonin transporter of rat cerebral cortical homogenate |
Bioorg Med Chem Lett 14: 5635-9 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.049
BindingDB Entry DOI: 10.7270/Q2RB743J |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50137997
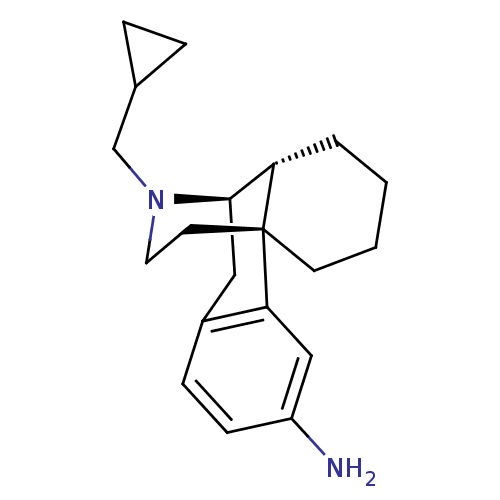
(3-Amino-N-cyclopropylmethylmorphinan | 3-aminocycl...)Show SMILES Nc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CC3)c2c1 Show InChI InChI=1S/C20H28N2/c21-16-7-6-15-11-19-17-3-1-2-8-20(17,18(15)12-16)9-10-22(19)13-14-4-5-14/h6-7,12,14,17,19H,1-5,8-11,13,21H2/t17-,19+,20+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69,593 from human kappa opioid receptor expressed in CHO membrane |
Bioorg Med Chem 15: 4106-12 (2007)
Article DOI: 10.1016/j.bmc.2007.03.076
BindingDB Entry DOI: 10.7270/Q2D50MN1 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50135797

(CHEMBL146756 | di[17-cyclobutylmethyl-(1R,9R,10R)-...)Show SMILES O=C(Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1)\C=C\C(=O)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C46H58N2O4/c49-43(51-35-15-13-33-25-41-37-11-1-3-19-45(37,39(33)27-35)21-23-47(41)29-31-7-5-8-31)17-18-44(50)52-36-16-14-34-26-42-38-12-2-4-20-46(38,40(34)28-36)22-24-48(42)30-32-9-6-10-32/h13-18,27-28,31-32,37-38,41-42H,1-12,19-26,29-30H2/b18-17+/t37-,38-,41+,42+,45+,46+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50017233
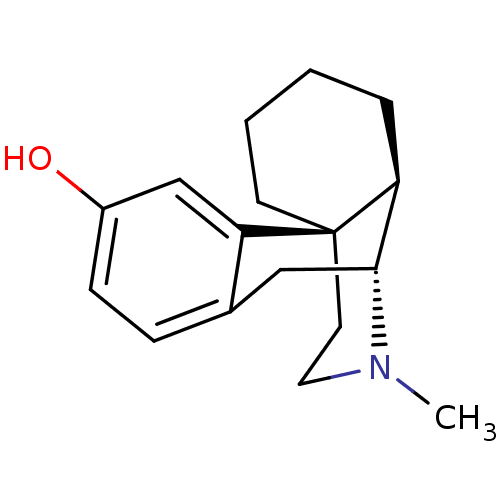
(CHEMBL592 | LEVO-DROMORAN | LEVORPHANOL)Show SMILES CN1CC[C@]23CCCC[C@H]2[C@H]1Cc1ccc(O)cc31 |r,TLB:0:1:12.18.11:9| Show InChI InChI=1S/C17H23NO/c1-18-9-8-17-7-3-2-4-14(17)16(18)10-12-5-6-13(19)11-15(12)17/h5-6,11,14,16,19H,2-4,7-10H2,1H3/t14-,16+,17+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50017233

(CHEMBL592 | LEVO-DROMORAN | LEVORPHANOL)Show SMILES CN1CC[C@]23CCCC[C@H]2[C@H]1Cc1ccc(O)cc31 |r,TLB:0:1:12.18.11:9| Show InChI InChI=1S/C17H23NO/c1-18-9-8-17-7-3-2-4-14(17)16(18)10-12-5-6-13(19)11-15(12)17/h5-6,11,14,16,19H,2-4,7-10H2,1H3/t14-,16+,17+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptors expressed in CHO cell membrane |
Bioorg Med Chem Lett 17: 1508-11 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.013
BindingDB Entry DOI: 10.7270/Q2WS8V2W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50180192

(1-((-)-N-cyclobutylmethylmorphinan-3-yl) 10-(3,6-d...)Show SMILES CN1CC[C@@]23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4O)ccc5OC(=O)CCCCCCCCC(=O)Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC3CCC3)c2c1 |c:16| Show InChI InChI=1S/C48H62N2O6/c1-49-25-24-48-36-19-20-40(51)46(48)56-45-41(21-17-33(44(45)48)28-38(36)49)55-43(53)15-7-5-3-2-4-6-14-42(52)54-34-18-16-32-27-39-35-13-8-9-22-47(35,37(32)29-34)23-26-50(39)30-31-11-10-12-31/h16-21,29,31,35-36,38-40,46,51H,2-15,22-28,30H2,1H3/t35?,36-,38+,39+,40-,46-,47+,48-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50135808

((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 |r,TLB:16:15:4.21.5:7| Show InChI InChI=1S/C21H29NO/c23-17-8-7-16-12-20-18-6-1-2-9-21(18,19(16)13-17)10-11-22(20)14-15-4-3-5-15/h7-8,13,15,18,20,23H,1-6,9-12,14H2/t18-,20+,21+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO membrane |
Bioorg Med Chem 15: 4106-12 (2007)
Article DOI: 10.1016/j.bmc.2007.03.076
BindingDB Entry DOI: 10.7270/Q2D50MN1 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50135808

((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 |r,TLB:16:15:4.21.5:7| Show InChI InChI=1S/C21H29NO/c23-17-8-7-16-12-20-18-6-1-2-9-21(18,19(16)13-17)10-11-22(20)14-15-4-3-5-15/h7-8,13,15,18,20,23H,1-6,9-12,14H2/t18-,20+,21+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Agonist activity at human opioid gamma receptor expressed in CHO cells assessed as inhibition of DAGO-stimulated [35S]GTPgammaS binding |
J Med Chem 50: 2254-8 (2007)
Article DOI: 10.1021/jm061327z
BindingDB Entry DOI: 10.7270/Q2CZ3804 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50135808

((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 |r,TLB:16:15:4.21.5:7| Show InChI InChI=1S/C21H29NO/c23-17-8-7-16-12-20-18-6-1-2-9-21(18,19(16)13-17)10-11-22(20)14-15-4-3-5-15/h7-8,13,15,18,20,23H,1-6,9-12,14H2/t18-,20+,21+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptors expressed in CHO cell membrane |
Bioorg Med Chem Lett 17: 1508-11 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.013
BindingDB Entry DOI: 10.7270/Q2WS8V2W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50180183
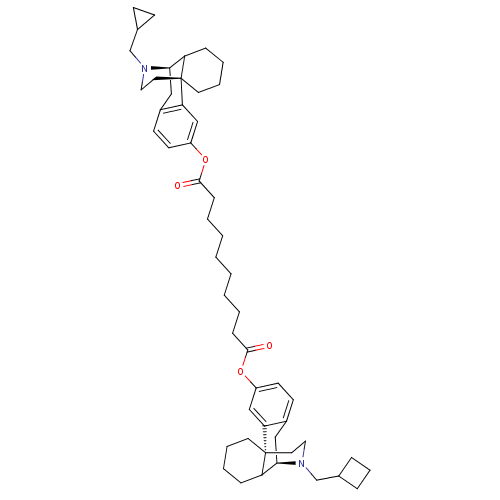
(1-((-)-N-cyclopropylmethylmorphinan-3-yl) 10-(N-cy...)Show SMILES O=C(CCCCCCCCC(=O)Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC3CCC3)c2c1)Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC3CC3)c2c1 Show InChI InChI=1S/C51H70N2O4/c54-48(56-40-22-20-38-30-46-42-14-7-9-24-50(42,44(38)32-40)26-28-52(46)34-36-12-11-13-36)16-5-3-1-2-4-6-17-49(55)57-41-23-21-39-31-47-43-15-8-10-25-51(43,45(39)33-41)27-29-53(47)35-37-18-19-37/h20-23,32-33,36-37,42-43,46-47H,1-19,24-31,34-35H2/t42?,43?,46-,47-,50-,51-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50135808

((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 |r,TLB:16:15:4.21.5:7| Show InChI InChI=1S/C21H29NO/c23-17-8-7-16-12-20-18-6-1-2-9-21(18,19(16)13-17)10-11-22(20)14-15-4-3-5-15/h7-8,13,15,18,20,23H,1-6,9-12,14H2/t18-,20+,21+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM60212

((4R,4aS,7aR,12bS)-3-(cyclopropylmethyl)-4a,9-bis(o...)Show SMILES Oc1ccc2C[C@H]3N(CC4CC4)CC[C@@]45[C@@H](Oc1c24)C(=O)CC[C@@]35O Show InChI InChI=1S/C20H23NO4/c22-13-4-3-12-9-15-20(24)6-5-14(23)18-19(20,16(12)17(13)25-18)7-8-21(15)10-11-1-2-11/h3-4,11,15,18,22,24H,1-2,5-10H2/t15-,18+,19+,20-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| DrugBank
Article
PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human opioid gamma receptor expressed in CHO cells |
J Med Chem 50: 2254-8 (2007)
Article DOI: 10.1021/jm061327z
BindingDB Entry DOI: 10.7270/Q2CZ3804 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Rattus norvegicus (rat)) | BDBM50155459
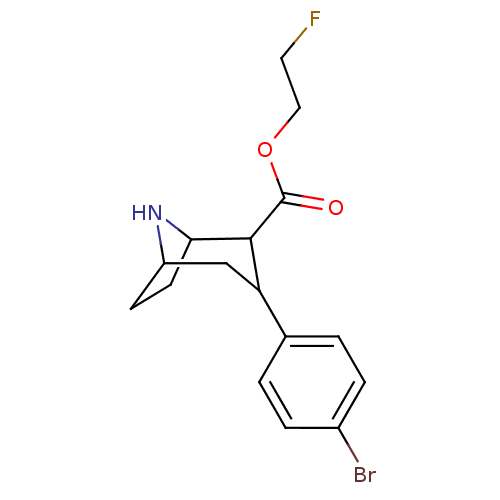
(3-(4-Bromo-phenyl)-8-aza-bicyclo[3.2.1]octane-2-ca...)Show SMILES FCCOC(=O)C1C2CCC(CC1c1ccc(Br)cc1)N2 |TLB:13:12:20:8.9,THB:4:6:20:8.9| Show InChI InChI=1S/C16H19BrFNO2/c17-11-3-1-10(2-4-11)13-9-12-5-6-14(19-12)15(13)16(20)21-8-7-18/h1-4,12-15,19H,5-9H2 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]-cyanoimiprimine from serotonin transporter of rat cerebral cortical homogenate |
Bioorg Med Chem Lett 14: 5635-9 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.049
BindingDB Entry DOI: 10.7270/Q2RB743J |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM60212

((4R,4aS,7aR,12bS)-3-(cyclopropylmethyl)-4a,9-bis(o...)Show SMILES Oc1ccc2C[C@H]3N(CC4CC4)CC[C@@]45[C@@H](Oc1c24)C(=O)CC[C@@]35O Show InChI InChI=1S/C20H23NO4/c22-13-4-3-12-9-15-20(24)6-5-14(23)18-19(20,16(12)17(13)25-18)7-8-21(15)10-11-1-2-11/h3-4,11,15,18,22,24H,1-2,5-10H2/t15-,18+,19+,20-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| DrugBank
Article
PubMed
| 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from human opioid kappa receptor expressed in CHO cells |
J Med Chem 50: 2254-8 (2007)
Article DOI: 10.1021/jm061327z
BindingDB Entry DOI: 10.7270/Q2CZ3804 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50180190

((1R,9R)-17-[(2S)-oxolan-2-ylmethyl]-17-azatetracyc...)Show SMILES Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3C[C@@H]3CCCO3)c2c1 |TLB:16:15:5.22.4:7| Show InChI InChI=1S/C21H29NO2/c23-16-7-6-15-12-20-18-5-1-2-8-21(18,19(15)13-16)9-10-22(20)14-17-4-3-11-24-17/h6-7,13,17-18,20,23H,1-5,8-12,14H2/t17-,18?,20+,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from human delta opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50194274
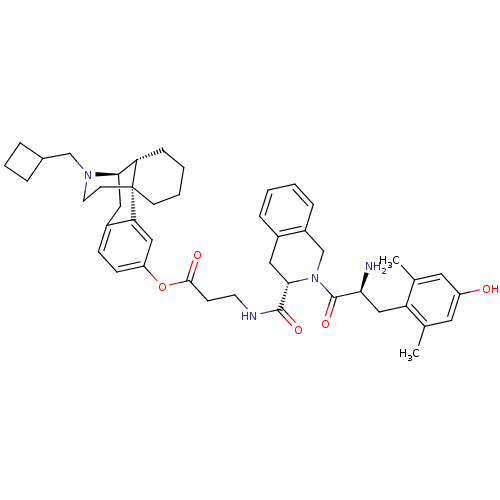
((-)-H-Dmt-Tic-beta-Ala-O-3-hydroxy-N-cyclobutylmet...)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@H]1C(=O)NCCC(=O)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C45H56N4O5/c1-28-20-34(50)21-29(2)36(28)25-39(46)44(53)49-27-33-11-4-3-10-31(33)22-41(49)43(52)47-18-15-42(51)54-35-14-13-32-23-40-37-12-5-6-16-45(37,38(32)24-35)17-19-48(40)26-30-8-7-9-30/h3-4,10-11,13-14,20-21,24,30,37,39-41,50H,5-9,12,15-19,22-23,25-27,46H2,1-2H3,(H,47,52)/t37-,39-,40+,41-,45+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells |
J Med Chem 49: 5640-3 (2006)
Article DOI: 10.1021/jm0605785
BindingDB Entry DOI: 10.7270/Q2CF9PQM |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50194274

((-)-H-Dmt-Tic-beta-Ala-O-3-hydroxy-N-cyclobutylmet...)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@H]1C(=O)NCCC(=O)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C45H56N4O5/c1-28-20-34(50)21-29(2)36(28)25-39(46)44(53)49-27-33-11-4-3-10-31(33)22-41(49)43(52)47-18-15-42(51)54-35-14-13-32-23-40-37-12-5-6-16-45(37,38(32)24-35)17-19-48(40)26-30-8-7-9-30/h3-4,10-11,13-14,20-21,24,30,37,39-41,50H,5-9,12,15-19,22-23,25-27,46H2,1-2H3,(H,47,52)/t37-,39-,40+,41-,45+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Inhibition of [3H]U-69593 binding to human kappa opioid receptor expressed in CHO cells at 10 uM |
J Med Chem 49: 5640-3 (2006)
Article DOI: 10.1021/jm0605785
BindingDB Entry DOI: 10.7270/Q2CF9PQM |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50204449

(CHEMBL427862 | MCL-449)Show SMILES CCNC(=O)Oc1ccc2CC3C4CCCCC4(CCN3CC3CC3)c2c1 |TLB:21:20:9.25.10:12,26:25:12:20.18.19| Show InChI InChI=1S/C23H32N2O2/c1-2-24-22(26)27-18-9-8-17-13-21-19-5-3-4-10-23(19,20(17)14-18)11-12-25(21)15-16-6-7-16/h8-9,14,16,19,21H,2-7,10-13,15H2,1H3,(H,24,26) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptors expressed in CHO cell membrane |
Bioorg Med Chem Lett 17: 1508-11 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.013
BindingDB Entry DOI: 10.7270/Q2WS8V2W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50180185
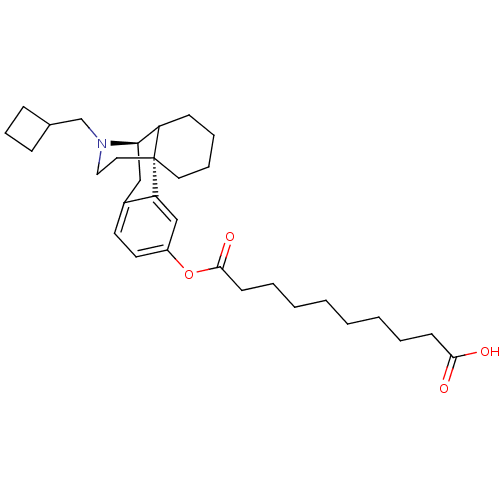
(10-{[(1R,9R)-17-(cyclobutylmethyl)-17-azatetracycl...)Show SMILES OC(=O)CCCCCCCCC(=O)Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C31H45NO4/c33-29(34)13-5-3-1-2-4-6-14-30(35)36-25-16-15-24-20-28-26-12-7-8-17-31(26,27(24)21-25)18-19-32(28)22-23-10-9-11-23/h15-16,21,23,26,28H,1-14,17-20,22H2,(H,33,34)/t26?,28-,31-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells |
J Med Chem 49: 256-62 (2006)
Article DOI: 10.1021/jm050577x
BindingDB Entry DOI: 10.7270/Q2HQ40QF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50209670

(17-(cyclopropylmethyl)morphinan-3-yl(5alpha)-17-(c...)Show SMILES O[C@]12CCC(=O)[C@@H]3Oc4c5c(C[C@H]1N(CC1CC1)CC[C@@]235)ccc4OC(=O)CCCCCCCCC(=O)Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC3CCC3)c2c1 |TLB:53:52:41.58.42:44| Show InChI InChI=1S/C51H66N2O7/c54-41-21-23-51(57)43-29-36-18-20-42(47-46(36)50(51,48(41)60-47)25-27-53(43)32-34-15-16-34)59-45(56)14-6-4-2-1-3-5-13-44(55)58-37-19-17-35-28-40-38-12-7-8-22-49(38,39(35)30-37)24-26-52(40)31-33-10-9-11-33/h17-20,30,33-34,38,40,43,48,57H,1-16,21-29,31-32H2/t38?,40-,43-,48+,49-,50+,51-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Agonist activity at human opioid gamma receptor expressed in CHO cells assessed as inhibition of DAGO-stimulated [35S]GTPgammaS binding |
J Med Chem 50: 2254-8 (2007)
Article DOI: 10.1021/jm061327z
BindingDB Entry DOI: 10.7270/Q2CZ3804 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50204452
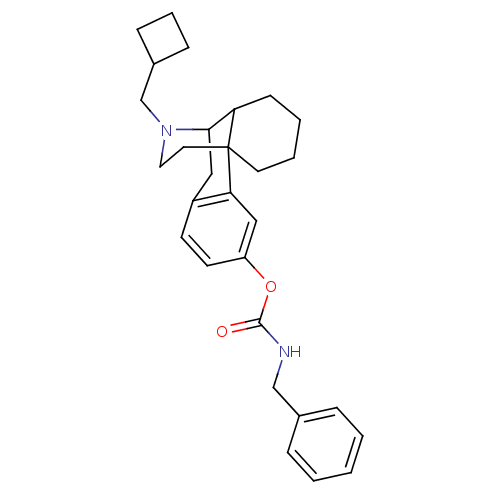
(CHEMBL394624 | MCL-443)Show SMILES O=C(NCc1ccccc1)Oc1ccc2CC3C4CCCCC4(CCN3CC3CCC3)c2c1 |TLB:26:25:14.31.15:17,32:31:17:25.23.24| Show InChI InChI=1S/C29H36N2O2/c32-28(30-19-21-7-2-1-3-8-21)33-24-13-12-23-17-27-25-11-4-5-14-29(25,26(23)18-24)15-16-31(27)20-22-9-6-10-22/h1-3,7-8,12-13,18,22,25,27H,4-6,9-11,14-17,19-20H2,(H,30,32) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from human kappa opioid receptors expressed in CHO cell membrane |
Bioorg Med Chem Lett 17: 1508-11 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.013
BindingDB Entry DOI: 10.7270/Q2WS8V2W |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data