| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Transthyretin |
|---|
| Ligand | BDBM50197883 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1670053 (CHEMBL4019941) |
|---|
| Kd | 278±n/a nM |
|---|
| Citation |  Smith, TP; Windsor, IW; Forest, KT; Raines, RT Stilbene Boronic Acids Form a Covalent Bond with Human Transthyretin and Inhibit Its Aggregation. J Med Chem60:7820-7834 (2017) [PubMed] Article Smith, TP; Windsor, IW; Forest, KT; Raines, RT Stilbene Boronic Acids Form a Covalent Bond with Human Transthyretin and Inhibit Its Aggregation. J Med Chem60:7820-7834 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Transthyretin |
|---|
| Name: | Transthyretin |
|---|
| Synonyms: | ATTR | PALB | Prealbumin | TBPA | TTHY_HUMAN | TTR | Transthyretin (TTR) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 15884.31 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P02766 |
|---|
| Residue: | 147 |
|---|
| Sequence: | MASHRLLLLCLAGLVFVSEAGPTGTGESKCPLMVKVLDAVRGSPAINVAVHVFRKAADDT
WEPFASGKTSESGELHGLTTEEEFVEGIYKVEIDTKSYWKALGISPFHEHAEVVFTANDS
GPRRYTIAALLSPYSYSTTAVVTNPKE
|
|
|
|---|
| BDBM50197883 |
|---|
| n/a |
|---|
| Name | BDBM50197883 |
|---|
| Synonyms: | CHEBI:78538 | FX-1006 | Tafamidis | US10377729, Compound Tafamidis | Vyndaqel |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H7Cl2NO3 |
|---|
| Mol. Mass. | 308.116 |
|---|
| SMILES | OC(=O)c1ccc2nc(oc2c1)-c1cc(Cl)cc(Cl)c1 |
|---|
| Structure | 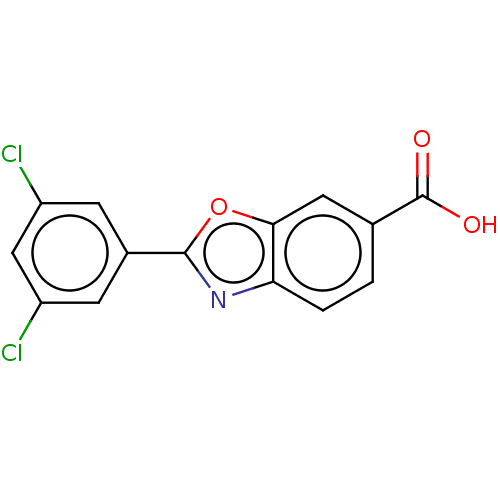
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Smith, TP; Windsor, IW; Forest, KT; Raines, RT Stilbene Boronic Acids Form a Covalent Bond with Human Transthyretin and Inhibit Its Aggregation. J Med Chem60:7820-7834 (2017) [PubMed] Article
Smith, TP; Windsor, IW; Forest, KT; Raines, RT Stilbene Boronic Acids Form a Covalent Bond with Human Transthyretin and Inhibit Its Aggregation. J Med Chem60:7820-7834 (2017) [PubMed] Article