| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50261088 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_1695601 |
|---|
| IC50 | >20000±n/a nM |
|---|
| Citation |  Wu, YJ; Guernon, J; McClure, A; Luo, G; Rajamani, R; Ng, A; Easton, A; Newton, A; Bourin, C; Parker, D; Mosure, K; Barnaby, O; Soars, MG; Knox, RJ; Matchett, M; Pieschl, R; Herrington, J; Chen, P; Sivarao, DV; Bristow, LJ; Meanwell, NA; Bronson, J; Olson, R; Thompson, LA; Dzierba, C Discovery of non-zwitterionic aryl sulfonamides as Na Bioorg Med Chem25:5490-5505 (2017) [PubMed] Article Wu, YJ; Guernon, J; McClure, A; Luo, G; Rajamani, R; Ng, A; Easton, A; Newton, A; Bourin, C; Parker, D; Mosure, K; Barnaby, O; Soars, MG; Knox, RJ; Matchett, M; Pieschl, R; Herrington, J; Chen, P; Sivarao, DV; Bristow, LJ; Meanwell, NA; Bronson, J; Olson, R; Thompson, LA; Dzierba, C Discovery of non-zwitterionic aryl sulfonamides as Na Bioorg Med Chem25:5490-5505 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50261088 |
|---|
| n/a |
|---|
| Name | BDBM50261088 |
|---|
| Synonyms: | CHEMBL4094490 | US10836758, Example 72 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H19ClFN3O3S2 |
|---|
| Mol. Mass. | 419.922 |
|---|
| SMILES | Fc1cc(NCCC2CCCOC2)c(Cl)cc1S(=O)(=O)Nc1nccs1 |
|---|
| Structure | 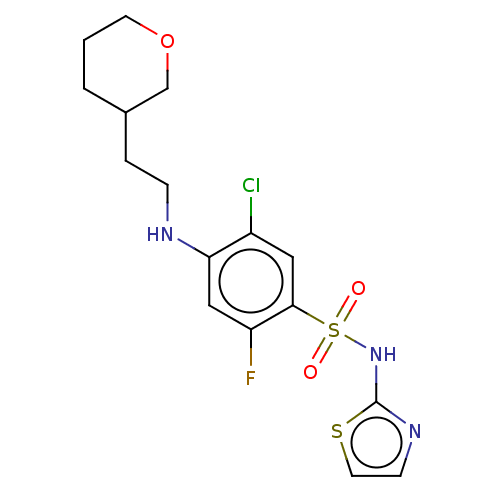
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wu, YJ; Guernon, J; McClure, A; Luo, G; Rajamani, R; Ng, A; Easton, A; Newton, A; Bourin, C; Parker, D; Mosure, K; Barnaby, O; Soars, MG; Knox, RJ; Matchett, M; Pieschl, R; Herrington, J; Chen, P; Sivarao, DV; Bristow, LJ; Meanwell, NA; Bronson, J; Olson, R; Thompson, LA; Dzierba, C Discovery of non-zwitterionic aryl sulfonamides as Na Bioorg Med Chem25:5490-5505 (2017) [PubMed] Article
Wu, YJ; Guernon, J; McClure, A; Luo, G; Rajamani, R; Ng, A; Easton, A; Newton, A; Bourin, C; Parker, D; Mosure, K; Barnaby, O; Soars, MG; Knox, RJ; Matchett, M; Pieschl, R; Herrington, J; Chen, P; Sivarao, DV; Bristow, LJ; Meanwell, NA; Bronson, J; Olson, R; Thompson, LA; Dzierba, C Discovery of non-zwitterionic aryl sulfonamides as Na Bioorg Med Chem25:5490-5505 (2017) [PubMed] Article