| Reaction Details |
|---|
 | Report a problem with these data |
| Target | C-C chemokine receptor type 5 |
|---|
| Ligand | BDBM50267125 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1702178 (CHEMBL4053411) |
|---|
| Kd | 10.0±n/a nM |
|---|
| Citation |  Schwehm, C; Kellam, B; Garces, AE; Hill, SJ; Kindon, ND; Bradshaw, TD; Li, J; Macdonald, SJ; Rowedder, JE; Stoddart, LA; Stocks, MJ Design and Elaboration of a Tractable Tricyclic Scaffold To Synthesize Druglike Inhibitors of Dipeptidyl Peptidase-4 (DPP-4), Antagonists of the C-C Chemokine Receptor Type 5 (CCR5), and Highly Potent and Selective Phosphoinositol-3 Kinase? (PI3K?) Inhibitors. J Med Chem60:1534-1554 (2017) [PubMed] Article Schwehm, C; Kellam, B; Garces, AE; Hill, SJ; Kindon, ND; Bradshaw, TD; Li, J; Macdonald, SJ; Rowedder, JE; Stoddart, LA; Stocks, MJ Design and Elaboration of a Tractable Tricyclic Scaffold To Synthesize Druglike Inhibitors of Dipeptidyl Peptidase-4 (DPP-4), Antagonists of the C-C Chemokine Receptor Type 5 (CCR5), and Highly Potent and Selective Phosphoinositol-3 Kinase? (PI3K?) Inhibitors. J Med Chem60:1534-1554 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| C-C chemokine receptor type 5 |
|---|
| Name: | C-C chemokine receptor type 5 |
|---|
| Synonyms: | C-C CKR-5 | C-C chemokine receptor type 5 | CC-CKR-5 | CCR-5 | CCR5 | CCR5/mu opioid receptor complex | CCR5_HUMAN | CD_antigen=CD195 | CHEMR13 | CMKBR5 | HIV-1 fusion coreceptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 40540.21 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P51681 |
|---|
| Residue: | 352 |
|---|
| Sequence: | MDYQVSSPIYDINYYTSEPCQKINVKQIAARLLPPLYSLVFIFGFVGNMLVILILINCKR
LKSMTDIYLLNLAISDLFFLLTVPFWAHYAAAQWDFGNTMCQLLTGLYFIGFFSGIFFII
LLTIDRYLAVVHAVFALKARTVTFGVVTSVITWVVAVFASLPGIIFTRSQKEGLHYTCSS
HFPYSQYQFWKNFQTLKIVILGLVLPLLVMVICYSGILKTLLRCRNEKKRHRAVRLIFTI
MIVYFLFWAPYNIVLLLNTFQEFFGLNNCSSSNRLDQAMQVTETLGMTHCCINPIIYAFV
GEKFRNYLLVFFQKHIAKRFCKCCSIFQQEAPERASSVYTRSTGEQEISVGL
|
|
|
|---|
| BDBM50267125 |
|---|
| n/a |
|---|
| Name | BDBM50267125 |
|---|
| Synonyms: | CHEMBL4089991 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H35F2N5O |
|---|
| Mol. Mass. | 471.5858 |
|---|
| SMILES | [H][C@@]12CCc3nnc(C)n3[C@]1([H])CCN(CC[C@H](NC(=O)C1CCC(F)(F)CC1)c1ccccc1)C2 |r| |
|---|
| Structure | 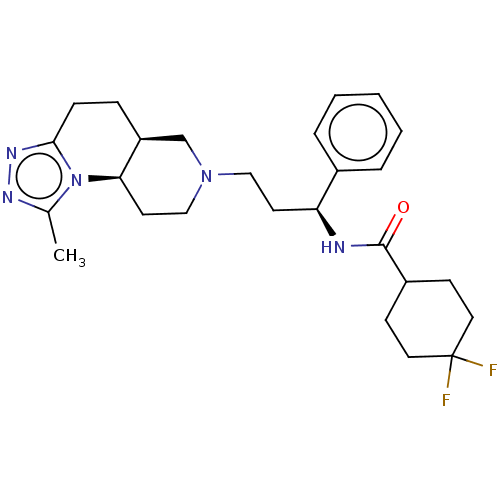
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Schwehm, C; Kellam, B; Garces, AE; Hill, SJ; Kindon, ND; Bradshaw, TD; Li, J; Macdonald, SJ; Rowedder, JE; Stoddart, LA; Stocks, MJ Design and Elaboration of a Tractable Tricyclic Scaffold To Synthesize Druglike Inhibitors of Dipeptidyl Peptidase-4 (DPP-4), Antagonists of the C-C Chemokine Receptor Type 5 (CCR5), and Highly Potent and Selective Phosphoinositol-3 Kinase? (PI3K?) Inhibitors. J Med Chem60:1534-1554 (2017) [PubMed] Article
Schwehm, C; Kellam, B; Garces, AE; Hill, SJ; Kindon, ND; Bradshaw, TD; Li, J; Macdonald, SJ; Rowedder, JE; Stoddart, LA; Stocks, MJ Design and Elaboration of a Tractable Tricyclic Scaffold To Synthesize Druglike Inhibitors of Dipeptidyl Peptidase-4 (DPP-4), Antagonists of the C-C Chemokine Receptor Type 5 (CCR5), and Highly Potent and Selective Phosphoinositol-3 Kinase? (PI3K?) Inhibitors. J Med Chem60:1534-1554 (2017) [PubMed] Article