| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Indoleamine 2,3-dioxygenase 1 |
|---|
| Ligand | BDBM73346 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1707498 (CHEMBL4058731) |
|---|
| IC50 | 3000±n/a nM |
|---|
| Citation |  Crosignani, S; Bingham, P; Bottemanne, P; Cannelle, H; Cauwenberghs, S; Cordonnier, M; Dalvie, D; Deroose, F; Feng, JL; Gomes, B; Greasley, S; Kaiser, SE; Kraus, M; N�grerie, M; Maegley, K; Miller, N; Murray, BW; Schneider, M; Soloweij, J; Stewart, AE; Tumang, J; Torti, VR; Van Den Eynde, B; Wythes, M Discovery of a Novel and Selective Indoleamine 2,3-Dioxygenase (IDO-1) Inhibitor 3-(5-Fluoro-1H-indol-3-yl)pyrrolidine-2,5-dione (EOS200271/PF-06840003) and Its Characterization as a Potential Clinical Candidate. J Med Chem60:9617-9629 (2017) [PubMed] Article Crosignani, S; Bingham, P; Bottemanne, P; Cannelle, H; Cauwenberghs, S; Cordonnier, M; Dalvie, D; Deroose, F; Feng, JL; Gomes, B; Greasley, S; Kaiser, SE; Kraus, M; N�grerie, M; Maegley, K; Miller, N; Murray, BW; Schneider, M; Soloweij, J; Stewart, AE; Tumang, J; Torti, VR; Van Den Eynde, B; Wythes, M Discovery of a Novel and Selective Indoleamine 2,3-Dioxygenase (IDO-1) Inhibitor 3-(5-Fluoro-1H-indol-3-yl)pyrrolidine-2,5-dione (EOS200271/PF-06840003) and Its Characterization as a Potential Clinical Candidate. J Med Chem60:9617-9629 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Indoleamine 2,3-dioxygenase 1 |
|---|
| Name: | Indoleamine 2,3-dioxygenase 1 |
|---|
| Synonyms: | I23O1_HUMAN | IDO | IDO-1 | IDO1 | INDO | Indoleamine 2,3-Dioxygenasae (IDO) | Indoleamine 2,3-dioxygenase | Indoleamine-pyrrole 2,3-dioxygenase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 45330.80 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P14902 |
|---|
| Residue: | 403 |
|---|
| Sequence: | MAHAMENSWTISKEYHIDEEVGFALPNPQENLPDFYNDWMFIAKHLPDLIESGQLRERVE
KLNMLSIDHLTDHKSQRLARLVLGCITMAYVWGKGHGDVRKVLPRNIAVPYCQLSKKLEL
PPILVYADCVLANWKKKDPNKPLTYENMDVLFSFRDGDCSKGFFLVSLLVEIAAASAIKV
IPTVFKAMQMQERDTLLKALLEIASCLEKALQVFHQIHDHVNPKAFFSVLRIYLSGWKGN
PQLSDGLVYEGFWEDPKEFAGGSAGQSSVFQCFDVLLGIQQTAGGGHAAQFLQDMRRYMP
PAHRNFLCSLESNPSVREFVLSKGDAGLREAYDACVKALVSLRSYHLQIVTKYILIPASQ
QPKENKTSEDPSKLEAKGTGGTDLMNFLKTVRSTTEKSLLKEG
|
|
|
|---|
| BDBM73346 |
|---|
| n/a |
|---|
| Name | BDBM73346 |
|---|
| Synonyms: | 3-(1H-indol-3-yl)-2,5-pyrrolidinedione | 3-(1H-indol-3-yl)pyrrolidine-2,5-dione | 3-(1H-indol-3-yl)pyrrolidine-2,5-quinone | MLS000099368 | SMR000071516 | US9603836, Compound 3 | US9949951, 3 | cid_363326 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C12H10N2O2 |
|---|
| Mol. Mass. | 214.22 |
|---|
| SMILES | O=C1CC(C(=O)N1)c1c[nH]c2ccccc12 |
|---|
| Structure | 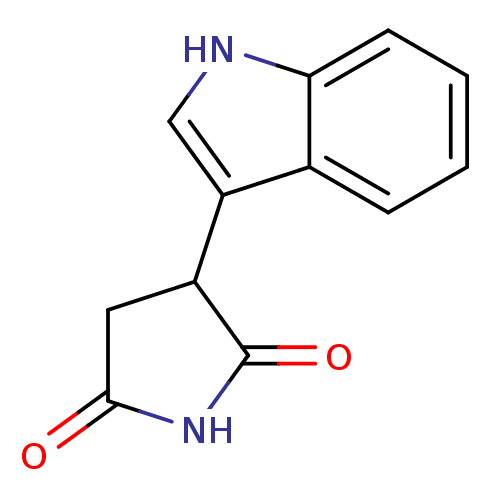
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Crosignani, S; Bingham, P; Bottemanne, P; Cannelle, H; Cauwenberghs, S; Cordonnier, M; Dalvie, D; Deroose, F; Feng, JL; Gomes, B; Greasley, S; Kaiser, SE; Kraus, M; N�grerie, M; Maegley, K; Miller, N; Murray, BW; Schneider, M; Soloweij, J; Stewart, AE; Tumang, J; Torti, VR; Van Den Eynde, B; Wythes, M Discovery of a Novel and Selective Indoleamine 2,3-Dioxygenase (IDO-1) Inhibitor 3-(5-Fluoro-1H-indol-3-yl)pyrrolidine-2,5-dione (EOS200271/PF-06840003) and Its Characterization as a Potential Clinical Candidate. J Med Chem60:9617-9629 (2017) [PubMed] Article
Crosignani, S; Bingham, P; Bottemanne, P; Cannelle, H; Cauwenberghs, S; Cordonnier, M; Dalvie, D; Deroose, F; Feng, JL; Gomes, B; Greasley, S; Kaiser, SE; Kraus, M; N�grerie, M; Maegley, K; Miller, N; Murray, BW; Schneider, M; Soloweij, J; Stewart, AE; Tumang, J; Torti, VR; Van Den Eynde, B; Wythes, M Discovery of a Novel and Selective Indoleamine 2,3-Dioxygenase (IDO-1) Inhibitor 3-(5-Fluoro-1H-indol-3-yl)pyrrolidine-2,5-dione (EOS200271/PF-06840003) and Its Characterization as a Potential Clinical Candidate. J Med Chem60:9617-9629 (2017) [PubMed] Article