| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cysteinyl leukotriene receptor 1 |
|---|
| Ligand | BDBM50013901 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_52379 |
|---|
| Ki | 11±n/a nM |
|---|
| Citation |  Galemmo, RA; Johnson, WH; Learn, KS; Lee, TD; Huang, FC; Campbell, HF; Youssefyeh, R; O'Rourke, SV; Schuessler, G; Sweeney, DM The development of a novel series of (quinolin-2-ylmethoxy)phenyl-containing compounds as high-affinity leukotriene receptor antagonists. 3. Structural variation of the acidic side chain to give antagonists of enhanced potency. J Med Chem33:2828-41 (1990) [PubMed] Galemmo, RA; Johnson, WH; Learn, KS; Lee, TD; Huang, FC; Campbell, HF; Youssefyeh, R; O'Rourke, SV; Schuessler, G; Sweeney, DM The development of a novel series of (quinolin-2-ylmethoxy)phenyl-containing compounds as high-affinity leukotriene receptor antagonists. 3. Structural variation of the acidic side chain to give antagonists of enhanced potency. J Med Chem33:2828-41 (1990) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cysteinyl leukotriene receptor 1 |
|---|
| Name: | Cysteinyl leukotriene receptor 1 |
|---|
| Synonyms: | CLTR1_HUMAN | CYSLT1 | CYSLTR1 | Cysteinyl leukotriene D4 receptor | Cysteinyl leukotriene receptor | Cysteinyl leukotriene receptor 1 | HG55 | HMTMF81 | LTD4 receptor | Leukotriene Cysteinyl 1 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 38565.16 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Leukotriene Cysteinyl 1 CYSLTR1 HUMAN::Q9Y271 |
|---|
| Residue: | 337 |
|---|
| Sequence: | MDETGNLTVSSATCHDTIDDFRNQVYSTLYSMISVVGFFGNGFVLYVLIKTYHKKSAFQV
YMINLAVADLLCVCTLPLRVVYYVHKGIWLFGDFLCRLSTYALYVNLYCSIFFMTAMSFF
RCIAIVFPVQNINLVTQKKARFVCVGIWIFVILTSSPFLMAKPQKDEKNNTKCFEPPQDN
QTKNHVLVLHYVSLFVGFIIPFVIIIVCYTMIILTLLKKSMKKNLSSHKKAIGMIMVVTA
AFLVSFMPYHIQRTIHLHFLHNETKPCDSVLRMQKSVVITLSLAASNCCFDPLLYFFSGG
NFRKRLSTFRKHSLSSVTYVPRKKASLPEKGEEICKV
|
|
|
|---|
| BDBM50013901 |
|---|
| n/a |
|---|
| Name | BDBM50013901 |
|---|
| Synonyms: | 2-(4-{2-[2-(1H-Tetrazol-5-yl)-ethyl]-pentyl}-phenoxymethyl)-quinoline | CHEMBL97356 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H27N5O |
|---|
| Mol. Mass. | 401.5041 |
|---|
| SMILES | CCCC(CCc1nnn[nH]1)Cc1ccc(OCc2ccc3ccccc3n2)cc1 |
|---|
| Structure | 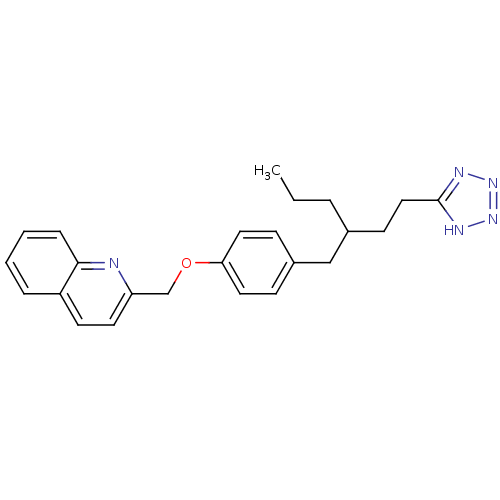
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Galemmo, RA; Johnson, WH; Learn, KS; Lee, TD; Huang, FC; Campbell, HF; Youssefyeh, R; O'Rourke, SV; Schuessler, G; Sweeney, DM The development of a novel series of (quinolin-2-ylmethoxy)phenyl-containing compounds as high-affinity leukotriene receptor antagonists. 3. Structural variation of the acidic side chain to give antagonists of enhanced potency. J Med Chem33:2828-41 (1990) [PubMed]
Galemmo, RA; Johnson, WH; Learn, KS; Lee, TD; Huang, FC; Campbell, HF; Youssefyeh, R; O'Rourke, SV; Schuessler, G; Sweeney, DM The development of a novel series of (quinolin-2-ylmethoxy)phenyl-containing compounds as high-affinity leukotriene receptor antagonists. 3. Structural variation of the acidic side chain to give antagonists of enhanced potency. J Med Chem33:2828-41 (1990) [PubMed]