| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM50271142 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1718383 (CHEMBL4133383) |
|---|
| IC50 | 0.270000±n/a nM |
|---|
| Citation |  Chierrito, TPC; Pedersoli-Mantoani, S; Roca, C; Requena, C; Sebastian-Perez, V; Castillo, WO; Moreira, NCS; P�rez, C; Sakamoto-Hojo, ET; Takahashi, CS; Jim�nez-Barbero, J; Ca�ada, FJ; Campillo, NE; Martinez, A; Carvalho, I From dual binding site acetylcholinesterase inhibitors to allosteric modulators: A new avenue for disease-modifying drugs in Alzheimer's disease. Eur J Med Chem139:773-791 (2017) [PubMed] Article Chierrito, TPC; Pedersoli-Mantoani, S; Roca, C; Requena, C; Sebastian-Perez, V; Castillo, WO; Moreira, NCS; P�rez, C; Sakamoto-Hojo, ET; Takahashi, CS; Jim�nez-Barbero, J; Ca�ada, FJ; Campillo, NE; Martinez, A; Carvalho, I From dual binding site acetylcholinesterase inhibitors to allosteric modulators: A new avenue for disease-modifying drugs in Alzheimer's disease. Eur J Med Chem139:773-791 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_HUMAN | ACHE | Acetylcholinesterase (AChE) | Acetylcholinesterase (human AChE) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 67792.70 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P22303 |
|---|
| Residue: | 614 |
|---|
| Sequence: | MRPPQCLLHTPSLASPLLLLLLWLLGGGVGAEGREDAELLVTVRGGRLRGIRLKTPGGPV
SAFLGIPFAEPPMGPRRFLPPEPKQPWSGVVDATTFQSVCYQYVDTLYPGFEGTEMWNPN
RELSEDCLYLNVWTPYPRPTSPTPVLVWIYGGGFYSGASSLDVYDGRFLVQAERTVLVSM
NYRVGAFGFLALPGSREAPGNVGLLDQRLALQWVQENVAAFGGDPTSVTLFGESAGAASV
GMHLLSPPSRGLFHRAVLQSGAPNGPWATVGMGEARRRATQLAHLVGCPPGGTGGNDTEL
VACLRTRPAQVLVNHEWHVLPQESVFRFSFVPVVDGDFLSDTPEALINAGDFHGLQVLVG
VVKDEGSYFLVYGAPGFSKDNESLISRAEFLAGVRVGVPQVSDLAAEAVVLHYTDWLHPE
DPARLREALSDVVGDHNVVCPVAQLAGRLAAQGARVYAYVFEHRASTLSWPLWMGVPHGY
EIEFIFGIPLDPSRNYTAEEKIFAQRLMRYWANFARTGDPNEPRDPKAPQWPPYTAGAQQ
YVSLDLRPLEVRRGLRAQACAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKN
QFDHYSKQDRCSDL
|
|
|
|---|
| BDBM50271142 |
|---|
| n/a |
|---|
| Name | BDBM50271142 |
|---|
| Synonyms: | 6-Chloro-9-[(3-{4-[(5,6-Dimethoxy-1-oxoindan-2-yl)methyl]piperidin-1-yl}propyl)amino]-1,2,3,4-tetrahydroacridine dihydrochloride | CHEMBL502725 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H40ClN3O3 |
|---|
| Mol. Mass. | 562.142 |
|---|
| SMILES | COc1cc2CC(CC3CCN(CCCNc4c5CCCCc5nc5cc(Cl)ccc45)CC3)C(=O)c2cc1OC |
|---|
| Structure | 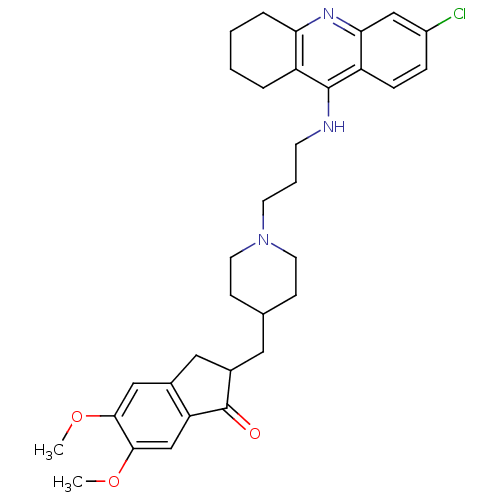
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Chierrito, TPC; Pedersoli-Mantoani, S; Roca, C; Requena, C; Sebastian-Perez, V; Castillo, WO; Moreira, NCS; P�rez, C; Sakamoto-Hojo, ET; Takahashi, CS; Jim�nez-Barbero, J; Ca�ada, FJ; Campillo, NE; Martinez, A; Carvalho, I From dual binding site acetylcholinesterase inhibitors to allosteric modulators: A new avenue for disease-modifying drugs in Alzheimer's disease. Eur J Med Chem139:773-791 (2017) [PubMed] Article
Chierrito, TPC; Pedersoli-Mantoani, S; Roca, C; Requena, C; Sebastian-Perez, V; Castillo, WO; Moreira, NCS; P�rez, C; Sakamoto-Hojo, ET; Takahashi, CS; Jim�nez-Barbero, J; Ca�ada, FJ; Campillo, NE; Martinez, A; Carvalho, I From dual binding site acetylcholinesterase inhibitors to allosteric modulators: A new avenue for disease-modifying drugs in Alzheimer's disease. Eur J Med Chem139:773-791 (2017) [PubMed] Article