| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Ectonucleoside triphosphate diphosphohydrolase 1 |
|---|
| Ligand | BDBM50468629 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1800816 (CHEMBL4273108) |
|---|
| IC50 | 10100±n/a nM |
|---|
| Citation |  Channar, PA; Afzal, S; Ejaz, SA; Saeed, A; Larik, FA; Mahesar, PA; Lecka, J; S�vigny, J; Erben, MF; Iqbal, J Exploration of carboxy pyrazole derivatives: Synthesis, alkaline phosphatase, nucleotide pyrophosphatase/phosphodiesterase and nucleoside triphosphate diphosphohydrolase inhibition studies with potential anticancer profile. Eur J Med Chem156:461-478 (2018) [PubMed] Article Channar, PA; Afzal, S; Ejaz, SA; Saeed, A; Larik, FA; Mahesar, PA; Lecka, J; S�vigny, J; Erben, MF; Iqbal, J Exploration of carboxy pyrazole derivatives: Synthesis, alkaline phosphatase, nucleotide pyrophosphatase/phosphodiesterase and nucleoside triphosphate diphosphohydrolase inhibition studies with potential anticancer profile. Eur J Med Chem156:461-478 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Ectonucleoside triphosphate diphosphohydrolase 1 |
|---|
| Name: | Ectonucleoside triphosphate diphosphohydrolase 1 |
|---|
| Synonyms: | ATPDase | CD_antigen=CD39 | Cd39 | ENTP1_RAT | Ecto-ATP diphosphohydrolase | Ecto-apyrase | Entpd1 | Lymphoid cell activation antigen | NTPDase 1 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 57412.31 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_463990 |
|---|
| Residue: | 511 |
|---|
| Sequence: | MEDIKDSKVKRFCSKNILIILGFSSVLAVIALIAVGLTHNKPLPENVKYGIVLDAGSSHT
NLYIYKWPAEKENDTGVVQLLEECQVKGPGISKYAQKTDEIAAYLAECMKMSTERIPASK
QHQTPVYLGATAGMRLLRMESKQSADEVLAAVSRSLKSYPFDFQGAKIITGQEEGAYGWI
TINYLLGRFTQEQSWLNFISDSQKQATFGALDLGGSSTQVTFVPLNQTLEAPETSLQFRL
YGTDYTVYTHSFLCYGKDQALWQKLAQDIQVSSGGILKDPCFYPGYKKVVNVSELYGTPC
TKRFEKKLPFNQFQVQGTGDYEQCHQSILKFFNNSHCPYSQCAFNGVFLPPLQGSFGAFS
AFYFVMDFFKKMANDSVSSQEKMTEITKNFCSKPWEEVKASYPTVKEKYLSEYCFSGTYI
LSLLLQGYNFTGTSWDQIHFMGKIKDSNAGWTLGYMLNLTNMIPAEQPLSPPLPHSTYIS
LMVLFSLVLVAMVITGLFIFSKPSYFWKEAV
|
|
|
|---|
| BDBM50468629 |
|---|
| n/a |
|---|
| Name | BDBM50468629 |
|---|
| Synonyms: | CHEMBL4279910 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C12H12N2O2 |
|---|
| Mol. Mass. | 216.2359 |
|---|
| SMILES | Cc1cc(=O)n([nH]1)C(=O)c1ccc(C)cc1 |
|---|
| Structure | 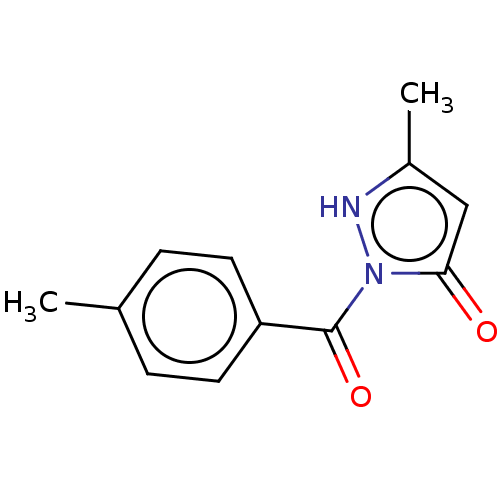
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Channar, PA; Afzal, S; Ejaz, SA; Saeed, A; Larik, FA; Mahesar, PA; Lecka, J; S�vigny, J; Erben, MF; Iqbal, J Exploration of carboxy pyrazole derivatives: Synthesis, alkaline phosphatase, nucleotide pyrophosphatase/phosphodiesterase and nucleoside triphosphate diphosphohydrolase inhibition studies with potential anticancer profile. Eur J Med Chem156:461-478 (2018) [PubMed] Article
Channar, PA; Afzal, S; Ejaz, SA; Saeed, A; Larik, FA; Mahesar, PA; Lecka, J; S�vigny, J; Erben, MF; Iqbal, J Exploration of carboxy pyrazole derivatives: Synthesis, alkaline phosphatase, nucleotide pyrophosphatase/phosphodiesterase and nucleoside triphosphate diphosphohydrolase inhibition studies with potential anticancer profile. Eur J Med Chem156:461-478 (2018) [PubMed] Article