| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histone acetyltransferase KAT2A |
|---|
| Ligand | BDBM50467835 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1802986 (CHEMBL4275278) |
|---|
| IC50 | 100000±n/a nM |
|---|
| Citation |  Zhang, R; Wang, J; Zhao, L; Liu, S; Du, D; Ding, H; Chen, S; Yue, L; Liu, YC; Zhang, C; Liu, H; Luo, C Identification of novel inhibitors of histone acetyltransferase hMOF through high throughput screening. Eur J Med Chem157:867-876 (2018) [PubMed] Article Zhang, R; Wang, J; Zhao, L; Liu, S; Du, D; Ding, H; Chen, S; Yue, L; Liu, YC; Zhang, C; Liu, H; Luo, C Identification of novel inhibitors of histone acetyltransferase hMOF through high throughput screening. Eur J Med Chem157:867-876 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histone acetyltransferase KAT2A |
|---|
| Name: | Histone acetyltransferase KAT2A |
|---|
| Synonyms: | GCN5 | GCN5 | GCN5L2 | General control of amino acid synthesis protein 5-like 2 | HGCN5 | Histone acetyltransferase GCN5 | Histone acetyltransferase KAT2A | Histone acetyltransferase KAT2A/KAT2B | HsGCN5 | KAT2A | KAT2A_HUMAN | Lysine acetyltransferase 2A | STAF97 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 93956.22 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_100876 |
|---|
| Residue: | 837 |
|---|
| Sequence: | MAEPSQAPTPAPAAQPRPLQSPAPAPTPTPAPSPASAPIPTPTPAPAPAPAAAPAGSTGT
GGPGVGSGGAGSGGDPARPGLSQQQRASQRKAQVRGLPRAKKLEKLGVFSACKANETCKC
NGWKNPKPPTAPRMDLQQPAANLSELCRSCEHPLADHVSHLENVSEDEINRLLGMVVDVE
NLFMSVHKEEDTDTKQVYFYLFKLLRKCILQMTRPVVEGSLGSPPFEKPNIEQGVLNFVQ
YKFSHLAPRERQTMFELSKMFLLCLNYWKLETPAQFRQRSQAEDVATYKVNYTRWLCYCH
VPQSCDSLPRYETTHVFGRSLLRSIFTVTRRQLLEKFRVEKDKLVPEKRTLILTHFPKFL
SMLEEEIYGANSPIWESGFTMPPSEGTQLVPRPASVSAAVVPSTPIFSPSMGGGSNSSLS
LDSAGAEPMPGEKRTLPENLTLEDAKRLRVMGDIPMELVNEVMLTITDPAAMLGPETSLL
SANAARDETARLEERRGIIEFHVIGNSLTPKANRRVLLWLVGLQNVFSHQLPRMPKEYIA
RLVFDPKHKTLALIKDGRVIGGICFRMFPTQGFTEIVFCAVTSNEQVKGYGTHLMNHLKE
YHIKHNILYFLTYADEYAIGYFKKQGFSKDIKVPKSRYLGYIKDYEGATLMECELNPRIP
YTELSHIIKKQKEIIKKLIERKQAQIRKVYPGLSCFKEGVRQIPVESVPGIRETGWKPLG
KEKGKELKDPDQLYTTLKNLLAQIKSHPSAWPFMEPVKKSEAPDYYEVIRFPIDLKTMTE
RLRSRYYVTRKLFVADLQRVIANCREYNPPDSEYCRCASALEKFFYFKLKEGGLIDK
|
|
|
|---|
| BDBM50467835 |
|---|
| n/a |
|---|
| Name | BDBM50467835 |
|---|
| Synonyms: | CHEMBL217676 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C9H12O4 |
|---|
| Mol. Mass. | 184.1892 |
|---|
| SMILES | CCC[C@H]1OC(=O)C(=C)[C@@H]1C(O)=O |r| |
|---|
| Structure | 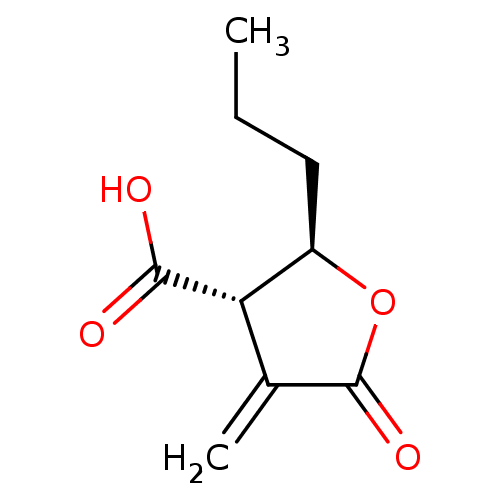
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhang, R; Wang, J; Zhao, L; Liu, S; Du, D; Ding, H; Chen, S; Yue, L; Liu, YC; Zhang, C; Liu, H; Luo, C Identification of novel inhibitors of histone acetyltransferase hMOF through high throughput screening. Eur J Med Chem157:867-876 (2018) [PubMed] Article
Zhang, R; Wang, J; Zhao, L; Liu, S; Du, D; Ding, H; Chen, S; Yue, L; Liu, YC; Zhang, C; Liu, H; Luo, C Identification of novel inhibitors of histone acetyltransferase hMOF through high throughput screening. Eur J Med Chem157:867-876 (2018) [PubMed] Article