| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM50321869 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2526 (CHEMBL616876) |
|---|
| Ki | 158±n/a nM |
|---|
| Citation |  Bromidge, SM; Dabbs, S; Davies, DT; Duckworth, DM; Forbes, IT; Ham, P; Jones, GE; King, FD; Saunders, DV; Starr, S; Thewlis, KM; Wyman, PA; Blaney, FE; Naylor, CB; Bailey, F; Blackburn, TP; Holland, V; Kennett, GA; Riley, GJ; Wood, MD Novel and selective 5-HT2C/2B receptor antagonists as potential anxiolytic agents: synthesis, quantitative structure-activity relationships, and molecular modeling of substituted 1-(3-pyridylcarbamoyl)indolines. J Med Chem41:1598-612 (1998) [PubMed] Article Bromidge, SM; Dabbs, S; Davies, DT; Duckworth, DM; Forbes, IT; Ham, P; Jones, GE; King, FD; Saunders, DV; Starr, S; Thewlis, KM; Wyman, PA; Blaney, FE; Naylor, CB; Bailey, F; Blackburn, TP; Holland, V; Kennett, GA; Riley, GJ; Wood, MD Novel and selective 5-HT2C/2B receptor antagonists as potential anxiolytic agents: synthesis, quantitative structure-activity relationships, and molecular modeling of substituted 1-(3-pyridylcarbamoyl)indolines. J Med Chem41:1598-612 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2 | 5-HT-2A | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT-2A) | 5-hydroxytryptamine receptor 2A (5HT-2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_HUMAN | HTR2 | HTR2A | Serotonin receptor 2A |
|---|
| Type: | undefined |
|---|
| Mol. Mass.: | 52607.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28223 |
|---|
| Residue: | 471 |
|---|
| Sequence: | MDILCEENTSLSSTTNSLMQLNDDTRLYSNDFNSGEANTSDAFNWTVDSENRTNLSCEGC
LSPSCLSLLHLQEKNWSALLTAVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAVWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VSFFIPLTIMVITYFLTIKSLQKEATLCVSDLGTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYTGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNEDVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENKKPLQLILVNTIPALAYK
SSQLQMGQKKNSKQDAKTTDNDCSMVALGKQHSEEASKDNSDGVNEKVSCV
|
|
|
|---|
| BDBM50321869 |
|---|
| n/a |
|---|
| Name | BDBM50321869 |
|---|
| Synonyms: | 6-Chloro-5-methyl-2,3-dihydro-indole-1-carboxylic acid pyridin-3-ylamide | 6-chloro-5-methyl-N-(pyridin-3-yl)indoline-1-carboxamide | CHEMBL42024 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H14ClN3O |
|---|
| Mol. Mass. | 287.744 |
|---|
| SMILES | Cc1cc2CCN(C(=O)Nc3cccnc3)c2cc1Cl |
|---|
| Structure | 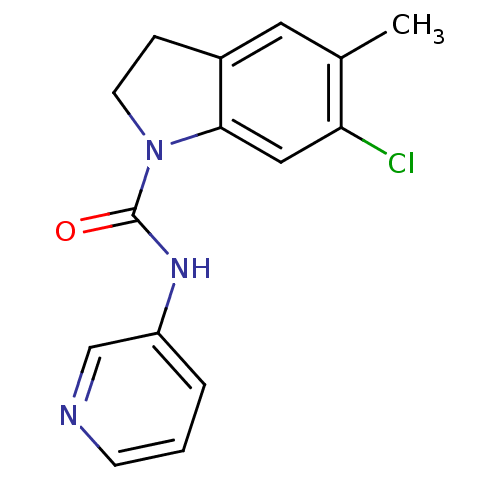
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bromidge, SM; Dabbs, S; Davies, DT; Duckworth, DM; Forbes, IT; Ham, P; Jones, GE; King, FD; Saunders, DV; Starr, S; Thewlis, KM; Wyman, PA; Blaney, FE; Naylor, CB; Bailey, F; Blackburn, TP; Holland, V; Kennett, GA; Riley, GJ; Wood, MD Novel and selective 5-HT2C/2B receptor antagonists as potential anxiolytic agents: synthesis, quantitative structure-activity relationships, and molecular modeling of substituted 1-(3-pyridylcarbamoyl)indolines. J Med Chem41:1598-612 (1998) [PubMed] Article
Bromidge, SM; Dabbs, S; Davies, DT; Duckworth, DM; Forbes, IT; Ham, P; Jones, GE; King, FD; Saunders, DV; Starr, S; Thewlis, KM; Wyman, PA; Blaney, FE; Naylor, CB; Bailey, F; Blackburn, TP; Holland, V; Kennett, GA; Riley, GJ; Wood, MD Novel and selective 5-HT2C/2B receptor antagonists as potential anxiolytic agents: synthesis, quantitative structure-activity relationships, and molecular modeling of substituted 1-(3-pyridylcarbamoyl)indolines. J Med Chem41:1598-612 (1998) [PubMed] Article