| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuronal acetylcholine receptor subunit alpha-7 |
|---|
| Ligand | BDBM50001942 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_144193 (CHEMBL750735) |
|---|
| Ki | 3.2±n/a nM |
|---|
| Citation |  Navarro, HA; Zhong, D; Abraham, P; Xu, H; Carroll, FI Synthesis and pharmacological characterization of [(125)I]iodomethyllycaconitine ([(125)I]iodo-MLA). A new ligand for the alpha(7) nicotinic acetylcholine receptor. J Med Chem43:142-5 (2000) [PubMed] Article Navarro, HA; Zhong, D; Abraham, P; Xu, H; Carroll, FI Synthesis and pharmacological characterization of [(125)I]iodomethyllycaconitine ([(125)I]iodo-MLA). A new ligand for the alpha(7) nicotinic acetylcholine receptor. J Med Chem43:142-5 (2000) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuronal acetylcholine receptor subunit alpha-7 |
|---|
| Name: | Neuronal acetylcholine receptor subunit alpha-7 |
|---|
| Synonyms: | ACHA7_RAT | Acra7 | Cholinergic, Nicotinic Alpha7 | Cholinergic, Nicotinic Alpha7/5-HT3 | Chrna7 | Neuronal acetylcholine receptor | Neuronal acetylcholine receptor (alpha7 nAChR) | Neuronal acetylcholine receptor subunit alpha 7 | Neuronal acetylcholine receptor subunit alpha-7 | Neuronal acetylcholine receptor subunit alpha-7 (nAChR alpha7) | Neuronal acetylcholine receptor subunit alpha-7 (nAChR) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 56502.44 |
|---|
| Organism: | Rattus norvegicus (Rat) |
|---|
| Description: | Q05941 |
|---|
| Residue: | 502 |
|---|
| Sequence: | MCGGRGGIWLALAAALLHVSLQGEFQRRLYKELVKNYNPLERPVANDSQPLTVYFSLSLL
QIMDVDEKNQVLTTNIWLQMSWTDHYLQWNMSEYPGVKNVRFPDGQIWKPDILLYNSADE
RFDATFHTNVLVNASGHCQYLPPGIFKSSCYIDVRWFPFDVQQCKLKFGSWSYGGWSLDL
QMQEADISSYIPNGEWDLMGIPGKRNEKFYECCKEPYPDVTYTVTMRRRTLYYGLNLLIP
CVLISALALLVFLLPADSGEKISLGITVLLSLTVFMLLVAEIMPATSDSVPLIAQYFAST
MIIVGLSVVVTVIVLRYHHHDPDGGKMPKWTRIILLNWCAWFLRMKRPGEDKVRPACQHK
PRRCSLASVELSAGAGPPTSNGNLLYIGFRGLEGMHCAPTPDSGVVCGRLACSPTHDEHL
MHGAHPSDGDPDLAKILEEVRYIANRFRCQDESEVICSEWKFAACVVDRLCLMAFSVFTI
ICTIGILMSAPNFVEAVSKDFA
|
|
|
|---|
| BDBM50001942 |
|---|
| n/a |
|---|
| Name | BDBM50001942 |
|---|
| Synonyms: | CHEMBL2113668 | Iodo-MLA |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C37H49IN2O10 |
|---|
| Mol. Mass. | 806.697 |
|---|
| SMILES | [H][C@]12C[C@]3([H])[C@]([H])([C@H]1OC)[C@](O)(C[C@@H]2OC)[C@]1(O)[C@@H](OC)[C@@]2([H])[C@@]33[C@H](CC[C@@]2(COC(=O)c2cc([125I])ccc2N2C(=O)C[C@H](C)C2=O)CN(CC)[C@@]13[H])OC |r,TLB:27:21:10.3.5:51,19:18:10.3.5:51,18:21:25.26.24:47.51.48,3:23:18.16:27.47.48,THB:24:23:18.16:27.47.48,8:7:3.2:10.13.12| |
|---|
| Structure | 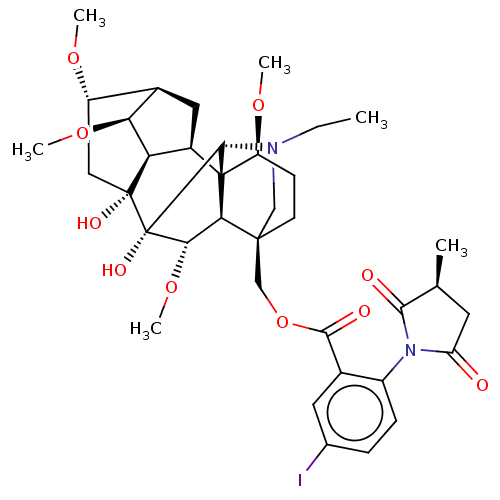
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Navarro, HA; Zhong, D; Abraham, P; Xu, H; Carroll, FI Synthesis and pharmacological characterization of [(125)I]iodomethyllycaconitine ([(125)I]iodo-MLA). A new ligand for the alpha(7) nicotinic acetylcholine receptor. J Med Chem43:142-5 (2000) [PubMed] Article
Navarro, HA; Zhong, D; Abraham, P; Xu, H; Carroll, FI Synthesis and pharmacological characterization of [(125)I]iodomethyllycaconitine ([(125)I]iodo-MLA). A new ligand for the alpha(7) nicotinic acetylcholine receptor. J Med Chem43:142-5 (2000) [PubMed] Article