| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Gastrin/cholecystokinin type B receptor |
|---|
| Ligand | BDBM81962 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_48758 (CHEMBL666784) |
|---|
| Ki | 269±n/a nM |
|---|
| Citation |  Ursini, A; Capelli, AM; Carr, RA; Cassar�, P; Corsi, M; Curcuruto, O; Curotto, G; Dal Cin, M; Davalli, S; Donati, D; Feriani, A; Finch, H; Finizia, G; Gaviraghi, G; Marien, M; Pentassuglia, G; Polinelli, S; Ratti, E; Reggiani, AM; Tarzia, G; Tedesco, G; Tranquillini, ME; Trist, DG; Van Amsterdam, FT; Reggiani, A Synthesis and SAR of new 5-phenyl-3-ureido-1,5-benzodiazepines as cholecystokinin-B receptor antagonists. J Med Chem43:3596-613 (2000) [PubMed] Article Ursini, A; Capelli, AM; Carr, RA; Cassar�, P; Corsi, M; Curcuruto, O; Curotto, G; Dal Cin, M; Davalli, S; Donati, D; Feriani, A; Finch, H; Finizia, G; Gaviraghi, G; Marien, M; Pentassuglia, G; Polinelli, S; Ratti, E; Reggiani, AM; Tarzia, G; Tedesco, G; Tranquillini, ME; Trist, DG; Van Amsterdam, FT; Reggiani, A Synthesis and SAR of new 5-phenyl-3-ureido-1,5-benzodiazepines as cholecystokinin-B receptor antagonists. J Med Chem43:3596-613 (2000) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Gastrin/cholecystokinin type B receptor |
|---|
| Name: | Gastrin/cholecystokinin type B receptor |
|---|
| Synonyms: | Cckbr | Cholecystokinin A | Cholecystokinin B receptor | Cholecystokinin receptor | GASR_RAT | Gastrin/cholecystokinin type B receptor |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 48980.43 |
|---|
| Organism: | RAT |
|---|
| Description: | Cholecystokinin A CCKBR RAT::P30553 |
|---|
| Residue: | 452 |
|---|
| Sequence: | MELLKLNRSVQGPGPGSGSSLCRPGVSLLNSSSAGNLSCDPPRIRGTGTRELEMAIRITL
YAVIFLMSVGGNVLIIVVLGLSRRLRTVTNAFLLSLAVSDLLLAVACMPFTLLPNLMGTF
IFGTVICKAISYLMGVSVSVSTLNLVAIALERYSAICRPLQARVWQTRSHAARVILATWL
LSGLLMVPYPVYTMVQPVGPRVLQCMHRWPSARVQQTWSVLLLLLLFFIPGVVIAVAYGL
ISRELYLGLHFDGENDSETQSRARNQGGLPGGAAPGPVHQNGGCRPVTSVAGEDSDGCCV
QLPRSRLEMTTLTTPTPGPVPGPRPNQAKLLAKKRVVRMLLVIVLLFFLCWLPVYSVNTW
RAFDGPGAQRALSGAPISFIHLLSYVSACVNPLVYCFMHRRFRQACLDTCARCCPRPPRA
RPQPLPDEDPPTPSIASLSRLSYTTISTLGPG
|
|
|
|---|
| BDBM81962 |
|---|
| n/a |
|---|
| Name | BDBM81962 |
|---|
| Synonyms: | S-L-365,260 |
|---|
| Type | n/a |
|---|
| Emp. Form. | C24H22N4O2 |
|---|
| Mol. Mass. | 398.4571 |
|---|
| SMILES | CN1c2ccccc2C(=N[C@@H](NC(=O)Nc2cccc(C)c2)C1=O)c1ccccc1 |r,c:9| |
|---|
| Structure | 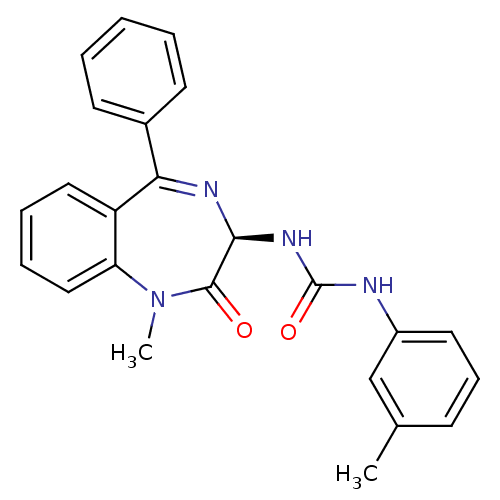
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ursini, A; Capelli, AM; Carr, RA; Cassar�, P; Corsi, M; Curcuruto, O; Curotto, G; Dal Cin, M; Davalli, S; Donati, D; Feriani, A; Finch, H; Finizia, G; Gaviraghi, G; Marien, M; Pentassuglia, G; Polinelli, S; Ratti, E; Reggiani, AM; Tarzia, G; Tedesco, G; Tranquillini, ME; Trist, DG; Van Amsterdam, FT; Reggiani, A Synthesis and SAR of new 5-phenyl-3-ureido-1,5-benzodiazepines as cholecystokinin-B receptor antagonists. J Med Chem43:3596-613 (2000) [PubMed] Article
Ursini, A; Capelli, AM; Carr, RA; Cassar�, P; Corsi, M; Curcuruto, O; Curotto, G; Dal Cin, M; Davalli, S; Donati, D; Feriani, A; Finch, H; Finizia, G; Gaviraghi, G; Marien, M; Pentassuglia, G; Polinelli, S; Ratti, E; Reggiani, AM; Tarzia, G; Tedesco, G; Tranquillini, ME; Trist, DG; Van Amsterdam, FT; Reggiani, A Synthesis and SAR of new 5-phenyl-3-ureido-1,5-benzodiazepines as cholecystokinin-B receptor antagonists. J Med Chem43:3596-613 (2000) [PubMed] Article