

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | 5-hydroxytryptamine receptor 1D | ||
| Ligand | BDBM50473505 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_1743 (CHEMBL616948) | ||
| IC50 | 7.1±n/a nM | ||
| Citation |  Jandu, KS; Barrett, V; Brockwell, M; Cambridge, D; Farrant, DR; Foster, C; Giles, H; Glen, RC; Hill, AP; Hobbs, H; Honey, A; Martin, GR; Salmon, J; Smith, D; Woollard, P; Selwood, DL Discovery of 4-[3-(trans-3-dimethylaminocyclobutyl)-1H-indol-5-ylmethyl]-(4S)-oxazolidin-2-one (4991W93), a 5HT(1B/1D) receptor partial agonist and a potent inhibitor of electrically induced plasma extravasation. J Med Chem44:681-93 (2001) [PubMed] Article Jandu, KS; Barrett, V; Brockwell, M; Cambridge, D; Farrant, DR; Foster, C; Giles, H; Glen, RC; Hill, AP; Hobbs, H; Honey, A; Martin, GR; Salmon, J; Smith, D; Woollard, P; Selwood, DL Discovery of 4-[3-(trans-3-dimethylaminocyclobutyl)-1H-indol-5-ylmethyl]-(4S)-oxazolidin-2-one (4991W93), a 5HT(1B/1D) receptor partial agonist and a potent inhibitor of electrically induced plasma extravasation. J Med Chem44:681-93 (2001) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| 5-hydroxytryptamine receptor 1D | |||
| Name: | 5-hydroxytryptamine receptor 1D | ||
| Synonyms: | 5-HT-1D | 5-HT-1D-alpha | 5-HT1D | 5-hydroxytryptamine receptor 1D (5-HT1D) | 5HT1D_HUMAN | HTR1D | HTR1DA | HTRL | Serotonin (5-HT) receptor | Serotonin Receptor 1D | ||
| Type: | G Protein-Coupled Receptor (GPCR) | ||
| Mol. Mass.: | 41920.63 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | Receptor binding assays were performed using human clone stably expressed in CHO cells. | ||
| Residue: | 377 | ||
| Sequence: |
| ||
| BDBM50473505 | |||
| n/a | |||
| Name | BDBM50473505 | ||
| Synonyms: | CHEMBL2368256 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C17H21N3O2 | ||
| Mol. Mass. | 299.3675 | ||
| SMILES | CN[C@H]1C[C@@H](C1)c1c[nH]c2ccc(CC3COC(=O)N3)cc12 |wU:2.1,wD:4.6,(17.04,-8.2,;15.54,-7.89,;15.06,-6.42,;13.68,-5.72,;14.37,-4.36,;15.75,-5.05,;13.89,-2.9,;14.79,-1.64,;13.87,-.4,;12.41,-.89,;11.07,-.12,;9.74,-.89,;9.74,-2.43,;8.41,-3.2,;8.41,-4.74,;7.17,-5.65,;7.64,-7.12,;9.18,-7.12,;10.27,-8.21,;9.66,-5.65,;11.07,-3.2,;12.42,-2.43,)| | ||
| Structure | 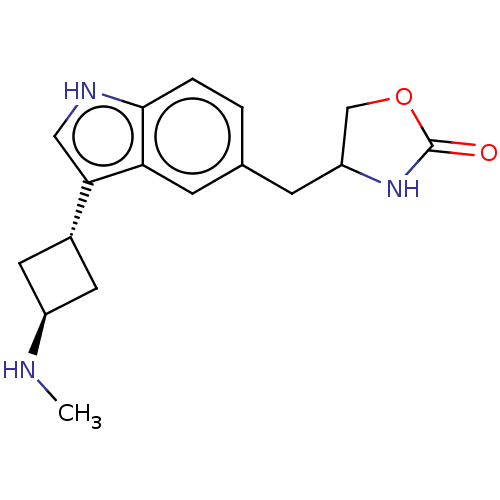
| ||