| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-1B adrenergic receptor |
|---|
| Ligand | BDBM50006952 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_425083 (CHEMBL912602) |
|---|
| Ki | 1.8±n/a nM |
|---|
| Citation |  Romeo, G; Materia, L; Marucci, G; Modica, M; Pittal�, V; Salerno, L; Siracusa, MA; Buccioni, M; Angeli, P; Minneman, KP New pyrimido[5,4-b]indoles and [1]benzothieno[3,2-d]pyrimidines: high affinity ligands for the alpha(1)-adrenoceptor subtypes. Bioorg Med Chem Lett16:6200-3 (2006) [PubMed] Article Romeo, G; Materia, L; Marucci, G; Modica, M; Pittal�, V; Salerno, L; Siracusa, MA; Buccioni, M; Angeli, P; Minneman, KP New pyrimido[5,4-b]indoles and [1]benzothieno[3,2-d]pyrimidines: high affinity ligands for the alpha(1)-adrenoceptor subtypes. Bioorg Med Chem Lett16:6200-3 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-1B adrenergic receptor |
|---|
| Name: | Alpha-1B adrenergic receptor |
|---|
| Synonyms: | ADA1B_HUMAN | ADRA1B | Adrenergic alpha1B | Adrenergic receptor | Adrenergic receptor alpha | Alpha 1B-adrenoceptor | Alpha 1B-adrenoreceptor | Alpha-1B adrenergic receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 56862.13 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P35368 |
|---|
| Residue: | 520 |
|---|
| Sequence: | MNPDLDTGHNTSAPAHWGELKNANFTGPNQTSSNSTLPQLDITRAISVGLVLGAFILFAI
VGNILVILSVACNRHLRTPTNYFIVNLAMADLLLSFTVLPFSAALEVLGYWVLGRIFCDI
WAAVDVLCCTASILSLCAISIDRYIGVRYSLQYPTLVTRRKAILALLSVWVLSTVISIGP
LLGWKEPAPNDDKECGVTEEPFYALFSSLGSFYIPLAVILVMYCRVYIVAKRTTKNLEAG
VMKEMSNSKELTLRIHSKNFHEDTLSSTKAKGHNPRSSIAVKLFKFSREKKAAKTLGIVV
GMFILCWLPFFIALPLGSLFSTLKPPDAVFKVVFWLGYFNSCLNPIIYPCSSKEFKRAFV
RILGCQCRGRGRRRRRRRRRLGGCAYTYRPWTRGGSLERSQSRKDSLDDSGSCLSGSQRT
LPSASPSPGYLGRGAPPPVELCAFPEWKAPGALLSLPAPEPPGRRGRHDSGPLFTFKLLT
EPESPGTDGGASNGGCEAAADVANGQPGFKSNMPLAPGQF
|
|
|
|---|
| BDBM50006952 |
|---|
| n/a |
|---|
| Name | BDBM50006952 |
|---|
| Synonyms: | 2-{2-[4-(2-Methoxy-phenyl)-piperazin-1-yl]-ethyl}-4,9-dihydro-2,4,9-triaza-fluorene-1,3-dione | 3-(2-(4-(2-methoxyphenyl)piperazin-1-yl)ethyl)-1H-pyrimido[5,4-b]indole-2,4(3H,5H)-dione | 3-{2-[4-(2-Methoxy-phenyl)-piperazin-1-yl]-ethyl}-1,5-dihydro-pyrimido[5,4-b]indole-2,4-dione | CHEMBL301707 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H25N5O3 |
|---|
| Mol. Mass. | 419.4763 |
|---|
| SMILES | COc1ccccc1N1CCN(CCn2c(=O)[nH]c3c4ccccc4[nH]c3c2=O)CC1 |
|---|
| Structure | 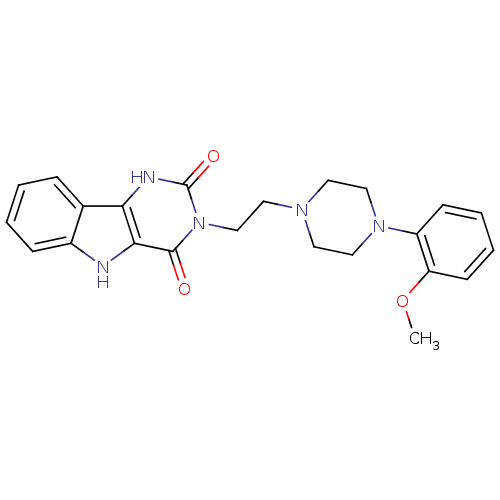
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Romeo, G; Materia, L; Marucci, G; Modica, M; Pittal�, V; Salerno, L; Siracusa, MA; Buccioni, M; Angeli, P; Minneman, KP New pyrimido[5,4-b]indoles and [1]benzothieno[3,2-d]pyrimidines: high affinity ligands for the alpha(1)-adrenoceptor subtypes. Bioorg Med Chem Lett16:6200-3 (2006) [PubMed] Article
Romeo, G; Materia, L; Marucci, G; Modica, M; Pittal�, V; Salerno, L; Siracusa, MA; Buccioni, M; Angeli, P; Minneman, KP New pyrimido[5,4-b]indoles and [1]benzothieno[3,2-d]pyrimidines: high affinity ligands for the alpha(1)-adrenoceptor subtypes. Bioorg Med Chem Lett16:6200-3 (2006) [PubMed] Article