| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM50029507 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_210582 (CHEMBL816532) |
|---|
| IC50 | 22±n/a nM |
|---|
| Citation |  Shuman, RT; Rothenberger, RB; Campbell, CS; Smith, GF; Gifford-Moore, DS; Paschal, JW; Gesellchen, PD Structure-activity study of tripeptide thrombin inhibitors using alpha-alkyl amino acids and other conformationally constrained amino acid substitutions. J Med Chem38:4446-53 (1995) [PubMed] Shuman, RT; Rothenberger, RB; Campbell, CS; Smith, GF; Gifford-Moore, DS; Paschal, JW; Gesellchen, PD Structure-activity study of tripeptide thrombin inhibitors using alpha-alkyl amino acids and other conformationally constrained amino acid substitutions. J Med Chem38:4446-53 (1995) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin | THRB_BOVIN | Thrombin | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 70502.73 |
|---|
| Organism: | Bos taurus (Bovine) |
|---|
| Description: | P00735 |
|---|
| Residue: | 625 |
|---|
| Sequence: | MARVRGPRLPGCLALAALFSLVHSQHVFLAHQQASSLLQRARRANKGFLEEVRKGNLERE
CLEEPCSREEAFEALESLSATDAFWAKYTACESARNPREKLNECLEGNCAEGVGMNYRGN
VSVTRSGIECQLWRSRYPHKPEINSTTHPGADLRENFCRNPDGSITGPWCYTTSPTLRRE
ECSVPVCGQDRVTVEVIPRSGGSTTSQSPLLETCVPDRGREYRGRLAVTTSGSRCLAWSS
EQAKALSKDQDFNPAVPLAENFCRNPDGDEEGAWCYVADQPGDFEYCDLNYCEEPVDGDL
GDRLGEDPDPDAAIEGRTSEDHFQPFFNEKTFGAGEADCGLRPLFEKKQVQDQTEKELFE
SYIEGRIVEGQDAEVGLSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNF
TVDDLLVRIGKHSRTRYERKVEKISMLDKIYIHPRYNWKENLDRDIALLKLKRPIELSDY
IHPVCLPDKQTAAKLLHAGFKGRVTGWGNRRETWTTSVAEVQPSVLQVVNLPLVERPVCK
ASTRIRITDNMFCAGYKPGEGKRGDACEGDSGGPFVMKSPYNNRWYQMGIVSWGEGCDRD
GKYGFYTHVFRLKKWIQKVIDRLGS
|
|
|
|---|
| BDBM50029507 |
|---|
| n/a |
|---|
| Name | BDBM50029507 |
|---|
| Synonyms: | CHEMBL2370860 | {(R)-2-[2-((S)-1-Formyl-4-guanidino-butylcarbamoyl)-azetidin-1-yl]-1-methyl-2-oxo-1-phenyl-ethyl}-carbamic acid tert-butyl ester |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H36N6O5 |
|---|
| Mol. Mass. | 488.5798 |
|---|
| SMILES | [#6]C([#6])([#6])[#8]-[#6](=O)-[#7][C@@]([#6])([#6](=O)-[#7]-1-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6]=O)c1ccccc1 |
|---|
| Structure | 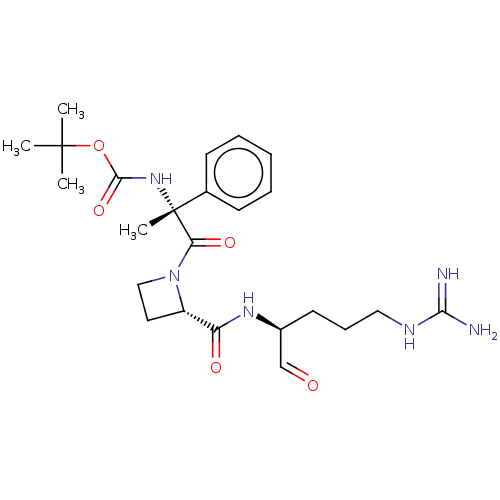
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Shuman, RT; Rothenberger, RB; Campbell, CS; Smith, GF; Gifford-Moore, DS; Paschal, JW; Gesellchen, PD Structure-activity study of tripeptide thrombin inhibitors using alpha-alkyl amino acids and other conformationally constrained amino acid substitutions. J Med Chem38:4446-53 (1995) [PubMed]
Shuman, RT; Rothenberger, RB; Campbell, CS; Smith, GF; Gifford-Moore, DS; Paschal, JW; Gesellchen, PD Structure-activity study of tripeptide thrombin inhibitors using alpha-alkyl amino acids and other conformationally constrained amino acid substitutions. J Med Chem38:4446-53 (1995) [PubMed]