| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Choline O-acetyltransferase |
|---|
| Ligand | BDBM50031187 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_27379 (CHEMBL642412) |
|---|
| Ki | >2000000±n/a nM |
|---|
| Citation |  Edwards, PD; Zottola, MA; Davis, M; Williams, J; Tuthill, PA Peptidyl alpha-ketoheterocyclic inhibitors of human neutrophil elastase. 3. In vitro and in vivo potency of a series of peptidyl alpha-ketobenzoxazoles. J Med Chem38:3972-82 (1995) [PubMed] Edwards, PD; Zottola, MA; Davis, M; Williams, J; Tuthill, PA Peptidyl alpha-ketoheterocyclic inhibitors of human neutrophil elastase. 3. In vitro and in vivo potency of a series of peptidyl alpha-ketobenzoxazoles. J Med Chem38:3972-82 (1995) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Choline O-acetyltransferase |
|---|
| Name: | Choline O-acetyltransferase |
|---|
| Synonyms: | CLAT_RAT | Chat | Choline Acetyltransferase | Choline O-acetyltransferase | Choline acetylase |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 71875.58 |
|---|
| Organism: | RAT |
|---|
| Description: | Choline Acetyltransferase 0 RAT::P32738 |
|---|
| Residue: | 640 |
|---|
| Sequence: | MPILEKAPQKMPVKASSWEELDLPKLPVPPLQQTLATYLQCMQHLVPEEQFRKSQAIVKR
FGAPGGLGETLQEKLLERQEKTANWVSEYWLNDMYLNNRLALPVNSSPAVIFARQHFQDT
NDQLRFAACLISGVLSYKTLLDSHSLPTDWAKGQLSGQPLCMKQYYRLFSSYRLPGHTQD
TLVAQKSSIMPEPEHVIVACCNQFFVLDVVINFRRLSEGDLFTQLRKIVKMASNEDERLP
PIGLLTSDGRSEWAKARTVLLKDSTNRDSLDMIERCICLVCLDGPGTGELSDTHRALQLL
HGGGCSLNGANRWYDKSLQFVVGRDGTCGVVCEHSPFDGIVLVQCTEHLLKHMMTSNKKL
VRADSVSELPAPRRLRLKCSPETQGHLASSAEKLQRIVKNLDFIVYKFDNYGKTFIKKQK
YSPDGFIQVALQLAYYRLYQRLVPTYESASIRRFQEGRVDNIRSATPEALAFVQAMTDHK
AAMPASEKLQLLQTAMQAHKQYTVMAITGMAIDNHLLALRELARDLCKEPPEMFMDETYL
MSNRFVLSTSQVPTTMEMFCCYGPVVPNGNGACYNPQPEAITFCISSFHSCKETSSVEFA
EAVGASLVDMRDLCSSRQPADSKPPAPKEKARGPSQAKQS
|
|
|
|---|
| BDBM50031187 |
|---|
| n/a |
|---|
| Name | BDBM50031187 |
|---|
| Synonyms: | 2-{(S)-2-[((S)-1-{(S)-2-[4-(4-Chloro-benzenesulfonylaminocarbonyl)-benzoylamino]-3-methyl-butyryl}-pyrrolidine-2-carbonyl)-amino]-3-methyl-butyryl}-benzooxazole-5-carboxylic acid amide | CHEMBL419985 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C37H39ClN6O9S |
|---|
| Mol. Mass. | 779.258 |
|---|
| SMILES | CC(C)[C@H](NC(=O)c1ccc(cc1)C(=O)NS(=O)(=O)c1ccc(Cl)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(=O)c1nc2cc(ccc2o1)C(N)=O |
|---|
| Structure | 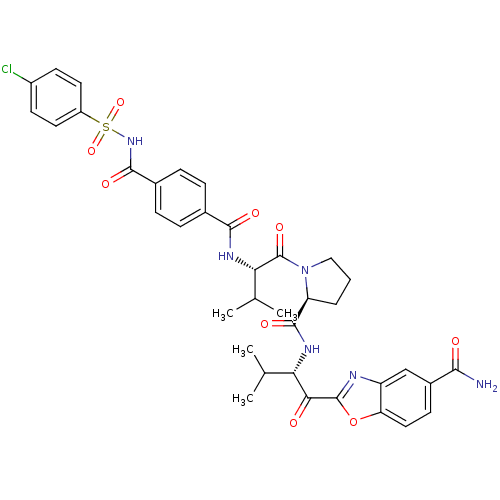
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Edwards, PD; Zottola, MA; Davis, M; Williams, J; Tuthill, PA Peptidyl alpha-ketoheterocyclic inhibitors of human neutrophil elastase. 3. In vitro and in vivo potency of a series of peptidyl alpha-ketobenzoxazoles. J Med Chem38:3972-82 (1995) [PubMed]
Edwards, PD; Zottola, MA; Davis, M; Williams, J; Tuthill, PA Peptidyl alpha-ketoheterocyclic inhibitors of human neutrophil elastase. 3. In vitro and in vivo potency of a series of peptidyl alpha-ketobenzoxazoles. J Med Chem38:3972-82 (1995) [PubMed]