| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Gastrin/cholecystokinin type B receptor |
|---|
| Ligand | BDBM50033636 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_47966 (CHEMBL653980) |
|---|
| IC50 | 1650±n/a nM |
|---|
| Citation |  Amblard, M; Rodriguez, M; Lignon, MF; Galas, MC; Bernad, N; Artis-Noël, AM; Hauad, L; Laur, J; Califano, JC; Aumelas, A Synthesis and biological evaluation of cholecystokinin analogs in which the Asp-Phe-NH2 moiety has been replaced by a 3-amino-7-phenylheptanoic acid or a 3-amino-6-(phenyloxy)hexanoic acid. J Med Chem36:3021-8 (1993) [PubMed] Amblard, M; Rodriguez, M; Lignon, MF; Galas, MC; Bernad, N; Artis-Noël, AM; Hauad, L; Laur, J; Califano, JC; Aumelas, A Synthesis and biological evaluation of cholecystokinin analogs in which the Asp-Phe-NH2 moiety has been replaced by a 3-amino-7-phenylheptanoic acid or a 3-amino-6-(phenyloxy)hexanoic acid. J Med Chem36:3021-8 (1993) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Gastrin/cholecystokinin type B receptor |
|---|
| Name: | Gastrin/cholecystokinin type B receptor |
|---|
| Synonyms: | CCK-2 receptor | CCK-B receptor | CCK-BR | CCKBR | CCKRB | Cholecystokinin A | Cholecystokinin receptor | Cholecystokinin-2 Receptor | GASR_HUMAN | Gastrin/cholecystokinin type B receptor |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 48445.79 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Stable expression of human CCK-2 receptors in HEK 293 cells. |
|---|
| Residue: | 447 |
|---|
| Sequence: | MELLKLNRSVQGTGPGPGASLCRPGAPLLNSSSVGNLSCEPPRIRGAGTRELELAIRITL
YAVIFLMSVGGNMLIIVVLGLSRRLRTVTNAFLLSLAVSDLLLAVACMPFTLLPNLMGTF
IFGTVICKAVSYLMGVSVSVSTLSLVAIALERYSAICRPLQARVWQTRSHAARVIVATWL
LSGLLMVPYPVYTVVQPVGPRVLQCVHRWPSARVRQTWSVLLLLLLFFIPGVVMAVAYGL
ISRELYLGLRFDGDSDSDSQSRVRNQGGLPGAVHQNGRCRPETGAVGEDSDGCYVQLPRS
RPALELTALTAPGPGSGSRPTQAKLLAKKRVVRMLLVIVVLFFLCWLPVYSANTWRAFDG
PGAHRALSGAPISFIHLLSYASACVNPLVYCFMHRRFRQACLETCARCCPRPPRARPRAL
PDEDPPTPSIASLSRLSYTTISTLGPG
|
|
|
|---|
| BDBM50033636 |
|---|
| n/a |
|---|
| Name | BDBM50033636 |
|---|
| Synonyms: | 3-{2-[2-(2-{2-[2-tert-Butoxycarbonylamino-3-(4-sulfo-phenyl)-propionylamino]-hexanoylamino}-acetylamino)-3-(1H-indol-3-yl)-propionylamino]-hexanoylamino}-6-phenoxy-hexanoic acid | CHEMBL385918 | CHEMBL414180 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C51H69N7O13S |
|---|
| Mol. Mass. | 1020.198 |
|---|
| SMILES | CCCC[C@H](NC(=O)[C@H](Cc1ccc(cc1)S(O)(=O)=O)NC(=O)OC(C)(C)C)C(=O)NCC(=O)N[C@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCC)C(=O)NC(CCCOc1ccccc1)CC(O)=O |
|---|
| Structure | 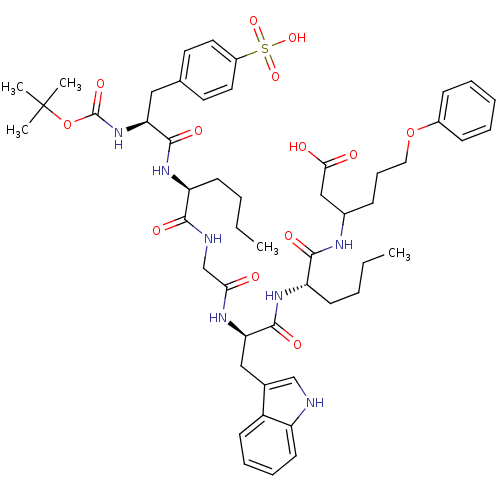
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Amblard, M; Rodriguez, M; Lignon, MF; Galas, MC; Bernad, N; Artis-Noël, AM; Hauad, L; Laur, J; Califano, JC; Aumelas, A Synthesis and biological evaluation of cholecystokinin analogs in which the Asp-Phe-NH2 moiety has been replaced by a 3-amino-7-phenylheptanoic acid or a 3-amino-6-(phenyloxy)hexanoic acid. J Med Chem36:3021-8 (1993) [PubMed]
Amblard, M; Rodriguez, M; Lignon, MF; Galas, MC; Bernad, N; Artis-Noël, AM; Hauad, L; Laur, J; Califano, JC; Aumelas, A Synthesis and biological evaluation of cholecystokinin analogs in which the Asp-Phe-NH2 moiety has been replaced by a 3-amino-7-phenylheptanoic acid or a 3-amino-6-(phenyloxy)hexanoic acid. J Med Chem36:3021-8 (1993) [PubMed]