| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A1 |
|---|
| Ligand | BDBM50011300 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_967978 (CHEMBL2401082) |
|---|
| Ki | 34±n/a nM |
|---|
| Citation |  Piersanti, G; Bartoccini, F; Lucarini, S; Cabri, W; Stasi, MA; Riccioni, T; Borsini, F; Tarzia, G; Minetti, P Synthesis and biological evaluation of metabolites of 2-n-butyl-9-methyl-8-[1,2,3]triazol-2-yl-9H-purin-6-ylamine (ST1535), a potent antagonist of the A2A adenosine receptor for the treatment of Parkinson's disease. J Med Chem56:5456-63 (2013) [PubMed] Article Piersanti, G; Bartoccini, F; Lucarini, S; Cabri, W; Stasi, MA; Riccioni, T; Borsini, F; Tarzia, G; Minetti, P Synthesis and biological evaluation of metabolites of 2-n-butyl-9-methyl-8-[1,2,3]triazol-2-yl-9H-purin-6-ylamine (ST1535), a potent antagonist of the A2A adenosine receptor for the treatment of Parkinson's disease. J Med Chem56:5456-63 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A1 |
|---|
| Name: | Adenosine receptor A1 |
|---|
| Synonyms: | A1 adenosine receptor (hA1) | A1AR | AA1R_HUMAN | ADENOSINE A1 | ADORA1 | Adenosine A1 receptor (A1AR) | Adenosine A1-receptor | Adenosine receptor A1 (A1) | Adenosine receptor A1 (hA1) | Adenosine transporter (AdT) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 36520.92 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P30542 |
|---|
| Residue: | 326 |
|---|
| Sequence: | MPPSISAFQAAYIGIEVLIALVSVPGNVLVIWAVKVNQALRDATFCFIVSLAVADVAVGA
LVIPLAILINIGPQTYFHTCLMVACPVLILTQSSILALLAIAVDRYLRVKIPLRYKMVVT
PRRAAVAIAGCWILSFVVGLTPMFGWNNLSAVERAWAANGSMGEPVIKCEFEKVISMEYM
VYFNFFVWVLPPLLLMVLIYLEVFYLIRKQLNKKVSASSGDPQKYYGKELKIAKSLALIL
FLFALSWLPLHILNCITLFCPSCHKPSILTYIAIFLTHGNSAMNPIVYAFRIQKFRVTFL
KIWNDHFRCQPAPPIDEDLPEERPDD
|
|
|
|---|
| BDBM50011300 |
|---|
| n/a |
|---|
| Name | BDBM50011300 |
|---|
| Synonyms: | CHEMBL2398482 | US9133197, 2 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C12H16N8O |
|---|
| Mol. Mass. | 288.3084 |
|---|
| SMILES | CC(O)CCc1nc(N)c2nc(-n3nccn3)n(C)c2n1 |
|---|
| Structure | 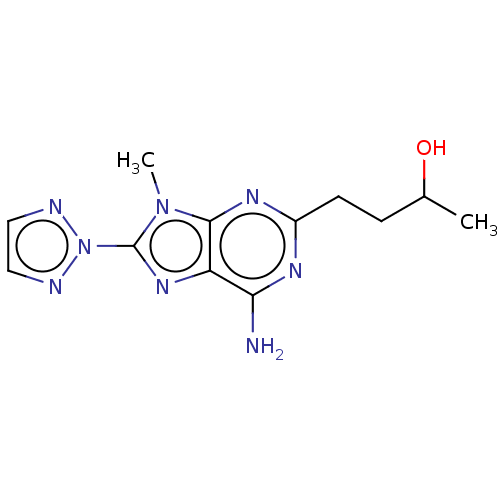
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Piersanti, G; Bartoccini, F; Lucarini, S; Cabri, W; Stasi, MA; Riccioni, T; Borsini, F; Tarzia, G; Minetti, P Synthesis and biological evaluation of metabolites of 2-n-butyl-9-methyl-8-[1,2,3]triazol-2-yl-9H-purin-6-ylamine (ST1535), a potent antagonist of the A2A adenosine receptor for the treatment of Parkinson's disease. J Med Chem56:5456-63 (2013) [PubMed] Article
Piersanti, G; Bartoccini, F; Lucarini, S; Cabri, W; Stasi, MA; Riccioni, T; Borsini, F; Tarzia, G; Minetti, P Synthesis and biological evaluation of metabolites of 2-n-butyl-9-methyl-8-[1,2,3]triazol-2-yl-9H-purin-6-ylamine (ST1535), a potent antagonist of the A2A adenosine receptor for the treatment of Parkinson's disease. J Med Chem56:5456-63 (2013) [PubMed] Article