| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin G/H synthase 1 |
|---|
| Ligand | BDBM50495258 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1284704 (CHEMBL3107404) |
|---|
| IC50 | 600±n/a nM |
|---|
| Citation |  Biava, M; Battilocchio, C; Poce, G; Alfonso, S; Consalvi, S; Di Capua, A; Calderone, V; Martelli, A; Testai, L; Sautebin, L; Rossi, A; Ghelardini, C; Di Cesare Mannelli, L; Giordani, A; Persiani, S; Colovic, M; Dovizio, M; Patrignani, P; Anzini, M Enhancing the pharmacodynamic profile of a class of selective COX-2 inhibiting nitric oxide donors. Bioorg Med Chem22:772-86 (2014) [PubMed] Article Biava, M; Battilocchio, C; Poce, G; Alfonso, S; Consalvi, S; Di Capua, A; Calderone, V; Martelli, A; Testai, L; Sautebin, L; Rossi, A; Ghelardini, C; Di Cesare Mannelli, L; Giordani, A; Persiani, S; Colovic, M; Dovizio, M; Patrignani, P; Anzini, M Enhancing the pharmacodynamic profile of a class of selective COX-2 inhibiting nitric oxide donors. Bioorg Med Chem22:772-86 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin G/H synthase 1 |
|---|
| Name: | Prostaglandin G/H synthase 1 |
|---|
| Synonyms: | COX-1 | COX1 | Cyclooxygenase | Cyclooxygenase-1 | Cyclooxygenase-1 (COX-1) | PGH1_HUMAN | PTGS1 | Prostaglandin G/H synthase (cyclooxygenase) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 68692.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P23219 |
|---|
| Residue: | 599 |
|---|
| Sequence: | MSRSLLLWFLLFLLLLPPLPVLLADPGAPTPVNPCCYYPCQHQGICVRFGLDRYQCDCTR
TGYSGPNCTIPGLWTWLRNSLRPSPSFTHFLLTHGRWFWEFVNATFIREMLMRLVLTVRS
NLIPSPPTYNSAHDYISWESFSNVSYYTRILPSVPKDCPTPMGTKGKKQLPDAQLLARRF
LLRRKFIPDPQGTNLMFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDNLERQ
YQLRLFKDGKLKYQVLDGEMYPPSVEEAPVLMHYPRGIPPQSQMAVGQEVFGLLPGLMLY
ATLWLREHNRVCDLLKAEHPTWGDEQLFQTTRLILIGETIKIVIEEYVQQLSGYFLQLKF
DPELLFGVQFQYRNRIAMEFNHLYHWHPLMPDSFKVGSQEYSYEQFLFNTSMLVDYGVEA
LVDAFSRQIAGRIGGGRNMDHHILHVAVDVIRESREMRLQPFNEYRKRFGMKPYTSFQEL
VGEKEMAAELEELYGDIDALEFYPGLLLEKCHPNSIFGESMIEIGAPFSLKGLLGNPICS
PEYWKPSTFGGEVGFNIVKTATLKKLVCLNTKTCPYVSFRVPDASQDDGPAVERPSTEL
|
|
|
|---|
| BDBM50495258 |
|---|
| n/a |
|---|
| Name | BDBM50495258 |
|---|
| Synonyms: | CHEMBL3104556 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H22FN3O6S |
|---|
| Mol. Mass. | 475.49 |
|---|
| SMILES | Cc1c(CC(=O)NCCO[N+]([O-])=O)cc(-c2ccc(cc2)S(C)(=O)=O)n1-c1ccc(F)cc1 |
|---|
| Structure | 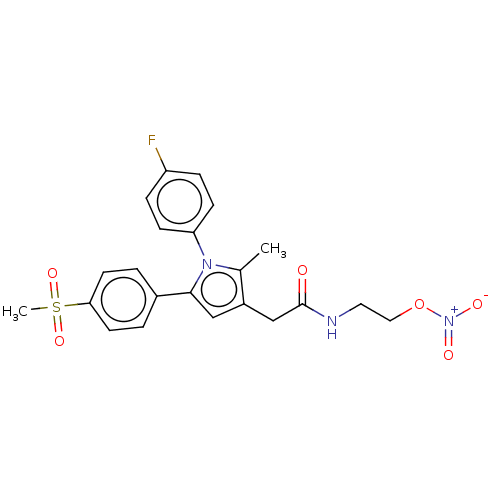
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Biava, M; Battilocchio, C; Poce, G; Alfonso, S; Consalvi, S; Di Capua, A; Calderone, V; Martelli, A; Testai, L; Sautebin, L; Rossi, A; Ghelardini, C; Di Cesare Mannelli, L; Giordani, A; Persiani, S; Colovic, M; Dovizio, M; Patrignani, P; Anzini, M Enhancing the pharmacodynamic profile of a class of selective COX-2 inhibiting nitric oxide donors. Bioorg Med Chem22:772-86 (2014) [PubMed] Article
Biava, M; Battilocchio, C; Poce, G; Alfonso, S; Consalvi, S; Di Capua, A; Calderone, V; Martelli, A; Testai, L; Sautebin, L; Rossi, A; Ghelardini, C; Di Cesare Mannelli, L; Giordani, A; Persiani, S; Colovic, M; Dovizio, M; Patrignani, P; Anzini, M Enhancing the pharmacodynamic profile of a class of selective COX-2 inhibiting nitric oxide donors. Bioorg Med Chem22:772-86 (2014) [PubMed] Article