| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Lutropin-choriogonadotropic hormone receptor |
|---|
| Ligand | BDBM50189778 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1328819 (CHEMBL3227403) |
|---|
| EC50 | 220±n/a nM |
|---|
| Citation |  Englund, EE; Neumann, S; Eliseeva, E; McCoy, JG; Titus, S; Zheng, W; Southall, N; Shin, P; Leister, W; Thomas, CJ; Inglese, J; Austin, CP; Gershengorn, MC; Huang, W The Synthesis and Evaluation of Dihydroquinazolin-4-ones and Quinazolin-4-ones as Thyroid Stimulating Hormone Receptor Agonists. Medchemcomm2:1016-1020 (2011) [PubMed] Article Englund, EE; Neumann, S; Eliseeva, E; McCoy, JG; Titus, S; Zheng, W; Southall, N; Shin, P; Leister, W; Thomas, CJ; Inglese, J; Austin, CP; Gershengorn, MC; Huang, W The Synthesis and Evaluation of Dihydroquinazolin-4-ones and Quinazolin-4-ones as Thyroid Stimulating Hormone Receptor Agonists. Medchemcomm2:1016-1020 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Lutropin-choriogonadotropic hormone receptor |
|---|
| Name: | Lutropin-choriogonadotropic hormone receptor |
|---|
| Synonyms: | LCGR | LGR2 | LH/CG-R | LHCGR | LHR | LHRHR | LSH-R | LSHR_HUMAN | Luteinizing hormone receptor |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 78662.77 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_562598 |
|---|
| Residue: | 699 |
|---|
| Sequence: | MKQRFSALQLLKLLLLLQPPLPRALREALCPEPCNCVPDGALRCPGPTAGLTRLSLAYLP
VKVIPSQAFRGLNEVIKIEISQIDSLERIEANAFDNLLNLSEILIQNTKNLRYIEPGAFI
NLPRLKYLSICNTGIRKFPDVTKVFSSESNFILEICDNLHITTIPGNAFQGMNNESVTLK
LYGNGFEEVQSHAFNGTTLTSLELKENVHLEKMHNGAFRGATGPKTLDISSTKLQALPSY
GLESIQRLIATSSYSLKKLPSRETFVNLLEATLTYPSHCCAFRNLPTKEQNFSHSISENF
SKQCESTVRKVNNKTLYSSMLAESELSGWDYEYGFCLPKTPRCAPEPDAFNPCEDIMGYD
FLRVLIWLINILAIMGNMTVLFVLLTSRYKLTVPRFLMCNLSFADFCMGLYLLLIASVDS
QTKGQYYNHAIDWQTGSGCSTAGFFTVFASELSVYTLTVITLERWHTITYAIHLDQKLRL
RHAILIMLGGWLFSSLIAMLPLVGVSNYMKVSICFPMDVETTLSQVYILTILILNVVAFF
IICACYIKIYFAVRNPELMATNKDTKIAKKMAILIFTDFTCMAPISFFAISAAFKVPLIT
VTNSKVLLVLFYPINSCANPFLYAIFTKTFQRDFFLLLSKFGCCKRRAELYRRKDFSAYT
SNCKNGFTGSNKPSQSTLKLSTLHCQGTALLDKTRYTEC
|
|
|
|---|
| BDBM50189778 |
|---|
| n/a |
|---|
| Name | BDBM50189778 |
|---|
| Synonyms: | CHEMBL211405 | N-tert-butyl-5-amino-4-(3-methoxyphenyl)-2-(methylthio)thieno[2,3-d]pyrimidine-6-carboxamide | ORG-41841 | cid_9887381 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H22N4O2S2 |
|---|
| Mol. Mass. | 402.534 |
|---|
| SMILES | COc1cccc(c1)-c1nc(SC)nc2sc(C(=O)NC(C)(C)C)c(N)c12 |
|---|
| Structure | 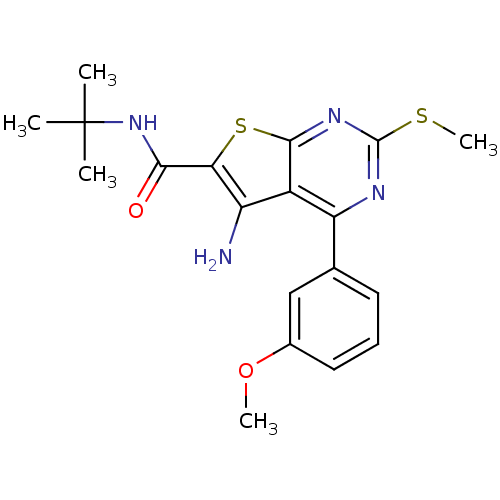
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Englund, EE; Neumann, S; Eliseeva, E; McCoy, JG; Titus, S; Zheng, W; Southall, N; Shin, P; Leister, W; Thomas, CJ; Inglese, J; Austin, CP; Gershengorn, MC; Huang, W The Synthesis and Evaluation of Dihydroquinazolin-4-ones and Quinazolin-4-ones as Thyroid Stimulating Hormone Receptor Agonists. Medchemcomm2:1016-1020 (2011) [PubMed] Article
Englund, EE; Neumann, S; Eliseeva, E; McCoy, JG; Titus, S; Zheng, W; Southall, N; Shin, P; Leister, W; Thomas, CJ; Inglese, J; Austin, CP; Gershengorn, MC; Huang, W The Synthesis and Evaluation of Dihydroquinazolin-4-ones and Quinazolin-4-ones as Thyroid Stimulating Hormone Receptor Agonists. Medchemcomm2:1016-1020 (2011) [PubMed] Article