| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 1,3-beta-D-glucan synthase catalytic subunit |
|---|
| Ligand | BDBM50499791 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1541190 (CHEMBL3745192) |
|---|
| IC50 | 1410±n/a nM |
|---|
| Citation |  Apgar, JM; Wilkening, RR; Greenlee, ML; Balkovec, JM; Flattery, AM; Abruzzo, GK; Galgoci, AM; Giacobbe, RA; Gill, CJ; Hsu, MJ; Liberator, P; Misura, AS; Motyl, M; Nielsen Kahn, J; Powles, M; Racine, F; Dragovic, J; Habulihaz, B; Fan, W; Kirwan, R; Lee, S; Liu, H; Mamai, A; Nelson, K; Peel, M Novel orally active inhibitors of ?-1,3-glucan synthesis derived from enfumafungin. Bioorg Med Chem Lett25:5813-8 (2015) [PubMed] Article Apgar, JM; Wilkening, RR; Greenlee, ML; Balkovec, JM; Flattery, AM; Abruzzo, GK; Galgoci, AM; Giacobbe, RA; Gill, CJ; Hsu, MJ; Liberator, P; Misura, AS; Motyl, M; Nielsen Kahn, J; Powles, M; Racine, F; Dragovic, J; Habulihaz, B; Fan, W; Kirwan, R; Lee, S; Liu, H; Mamai, A; Nelson, K; Peel, M Novel orally active inhibitors of ?-1,3-glucan synthesis derived from enfumafungin. Bioorg Med Chem Lett25:5813-8 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 1,3-beta-D-glucan synthase catalytic subunit |
|---|
| Name: | 1,3-beta-D-glucan synthase catalytic subunit |
|---|
| Synonyms: | 1,3-beta-D-glucan synthase catalytic subunit | Beta-1,3-glucan synthase |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 80469.77 |
|---|
| Organism: | Candida albicans |
|---|
| Description: | ChEMBL_663643 |
|---|
| Residue: | 690 |
|---|
| Sequence: | GTIYWMYTAYNSPTLYTKHYVQTINQQPLASSRWAACAIGGVLASFIQILATLFEWIFVP
REWAGAQHLSRRMLFLVLIFLLNLVPPVYTFQITKLVIYSKSAYAVSIVGFFIAVATLVF
FAVMPLGGLFTSYMNKRSRRYIASQTFTANYIKLKGLDMWMSYLLWFLVFLAKLVESYFF
LTLSLRDPIRNLSTMTMRCVGEVWYKDIVCRNQAKIVLGLMYLVDLLLFFLDTYMWYIIC
NCIFSIGRSFYLGISILTPWRNIFTRLPKRIYSKILATTEMEIKYKPKVLISQIWNAIVI
SMYREHLLAIDHVQKLLYHQVPSEIEGKRTLRAPTFFVSQDDNNFETEFFPRNSEAERRI
SFFAQSLATPMPEPLPVDNMPTFTVFTPHYSEKILLSLREIIREDDQFSRVTLLEYLKQL
HPVEWDCFVKDTKILAEETAAYENGDDSEKLSEDGLKSKIDDLPFYCIGFKSAAPEYTLR
TRIWASLRSQTLYRTVSGFMNYARAIKLLYRVENPELVQYFGGDPEGLELALERMARRKF
RFLVSMQRLSKFKDDEMENAEFLLRAYPDLQIAYLDEEPALNEDEEPRVYSALIDGHCEM
LENGRRRPKFRVQLSGNPILGDGKSDNQNHAVIFHRGEYIQLIDANQDNYLEECLKIRSV
LAEFEEMNVEHVNPYAPNLKSEDNNTKKDP
|
|
|
|---|
| BDBM50499791 |
|---|
| n/a |
|---|
| Name | BDBM50499791 |
|---|
| Synonyms: | CHEMBL3741042 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C38H59N3O9 |
|---|
| Mol. Mass. | 701.8898 |
|---|
| SMILES | [H][C@@]1(O[C@H]2[C@@H](C[C@@]34COC[C@]2(C)[C@]3([H])CC[C@@]2([H])C4=CC[C@@]3(C)[C@H](C(O)=O)[C@](C)(CC[C@]23C)[C@H](C)C(C)C)n2nccn2)O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O |r,t:21| |
|---|
| Structure | 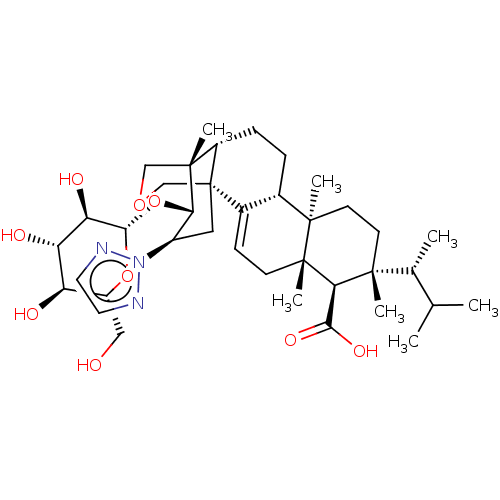
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Apgar, JM; Wilkening, RR; Greenlee, ML; Balkovec, JM; Flattery, AM; Abruzzo, GK; Galgoci, AM; Giacobbe, RA; Gill, CJ; Hsu, MJ; Liberator, P; Misura, AS; Motyl, M; Nielsen Kahn, J; Powles, M; Racine, F; Dragovic, J; Habulihaz, B; Fan, W; Kirwan, R; Lee, S; Liu, H; Mamai, A; Nelson, K; Peel, M Novel orally active inhibitors of ?-1,3-glucan synthesis derived from enfumafungin. Bioorg Med Chem Lett25:5813-8 (2015) [PubMed] Article
Apgar, JM; Wilkening, RR; Greenlee, ML; Balkovec, JM; Flattery, AM; Abruzzo, GK; Galgoci, AM; Giacobbe, RA; Gill, CJ; Hsu, MJ; Liberator, P; Misura, AS; Motyl, M; Nielsen Kahn, J; Powles, M; Racine, F; Dragovic, J; Habulihaz, B; Fan, W; Kirwan, R; Lee, S; Liu, H; Mamai, A; Nelson, K; Peel, M Novel orally active inhibitors of ?-1,3-glucan synthesis derived from enfumafungin. Bioorg Med Chem Lett25:5813-8 (2015) [PubMed] Article