| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM50279768 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_208499 (CHEMBL811974) |
|---|
| pH | 7.5±n/a |
|---|
| Ki | >15000±n/a nM |
|---|
| Comments | extracted |
|---|
| Citation |  Regan, J; McGarry, D; Bruno, J; Green, D; Newman, J; Hsu, CY; Kline, J; Barton, J; Travis, J; Choi, YM; Volz, F; Pauls, H; Harrison, R; Zilberstein, A; Ben-Sasson, SA; Chang, M Anionic- and lipophilic-mediated surface binding inhibitors of human leukocyte elastase. J Med Chem40:3408-22 (1997) [PubMed] Article Regan, J; McGarry, D; Bruno, J; Green, D; Newman, J; Hsu, CY; Kline, J; Barton, J; Travis, J; Choi, YM; Volz, F; Pauls, H; Harrison, R; Zilberstein, A; Ben-Sasson, SA; Chang, M Anionic- and lipophilic-mediated surface binding inhibitors of human leukocyte elastase. J Med Chem40:3408-22 (1997) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 70029.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00734 |
|---|
| Residue: | 622 |
|---|
| Sequence: | MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLEREC
VEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHV
NITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQE
CSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASA
QAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETG
DGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYI
DGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTEN
DLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHP
VCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDST
RIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKY
GFYTHVFRLKKWIQKVIDQFGE
|
|
|
|---|
| BDBM50279768 |
|---|
| n/a |
|---|
| Name | BDBM50279768 |
|---|
| Synonyms: | (2-{2-Benzyloxy-4-[2-benzyloxy-4-(2-benzyloxy-5-carboxymethoxy-4-hydroxymethyl-benzyl)-5-carboxymethoxy-benzyl]-5-carboxymethoxy-benzyl}-4-hydroxy-phenoxy)-acetic acid | CHEMBL216442 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C57H52O17 |
|---|
| Mol. Mass. | 1009.0126 |
|---|
| SMILES | OCc1cc(OCc2ccccc2)c(Cc2cc(OCc3ccccc3)c(Cc3cc(OCc4ccccc4)c(Cc4cc(O)ccc4OCC(O)=O)cc3OCC(O)=O)cc2OCC(O)=O)cc1OCC(O)=O |
|---|
| Structure | 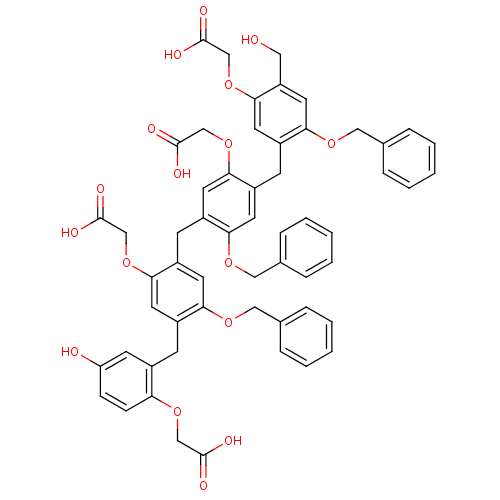
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Regan, J; McGarry, D; Bruno, J; Green, D; Newman, J; Hsu, CY; Kline, J; Barton, J; Travis, J; Choi, YM; Volz, F; Pauls, H; Harrison, R; Zilberstein, A; Ben-Sasson, SA; Chang, M Anionic- and lipophilic-mediated surface binding inhibitors of human leukocyte elastase. J Med Chem40:3408-22 (1997) [PubMed] Article
Regan, J; McGarry, D; Bruno, J; Green, D; Newman, J; Hsu, CY; Kline, J; Barton, J; Travis, J; Choi, YM; Volz, F; Pauls, H; Harrison, R; Zilberstein, A; Ben-Sasson, SA; Chang, M Anionic- and lipophilic-mediated surface binding inhibitors of human leukocyte elastase. J Med Chem40:3408-22 (1997) [PubMed] Article