| Reaction Details |
|---|
 | Report a problem with these data |
| Target | C-C chemokine receptor type 7 |
|---|
| Ligand | BDBM50088301 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1843869 (CHEMBL4344296) |
|---|
| IC50 | 1.4±n/a nM |
|---|
| Citation |  Romero, FA; Jones, CT; Xu, Y; Fenaux, M; Halcomb, RL The Race to Bash NASH: Emerging Targets and Drug Development in a Complex Liver Disease. J Med Chem63:5031-5073 (2020) [PubMed] Article Romero, FA; Jones, CT; Xu, Y; Fenaux, M; Halcomb, RL The Race to Bash NASH: Emerging Targets and Drug Development in a Complex Liver Disease. J Med Chem63:5031-5073 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| C-C chemokine receptor type 7 |
|---|
| Name: | C-C chemokine receptor type 7 |
|---|
| Synonyms: | CCR7 | CCR7_HUMAN | CMKBR7 | EBI1 | EVI1 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 42885.12 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_338819 |
|---|
| Residue: | 378 |
|---|
| Sequence: | MDLGKPMKSVLVVALLVIFQVCLCQDEVTDDYIGDNTTVDYTLFESLCSKKDVRNFKAWF
LPIMYSIICFVGLLGNGLVVLTYIYFKRLKTMTDTYLLNLAVADILFLLTLPFWAYSAAK
SWVFGVHFCKLIFAIYKMSFFSGMLLLLCISIDRYVAIVQAVSAHRHRARVLLISKLSCV
GIWILATVLSIPELLYSDLQRSSSEQAMRCSLITEHVEAFITIQVAQMVIGFLVPLLAMS
FCYLVIIRTLLQARNFERNKAIKVIIAVVVVFIVFQLPYNGVVLAQTVANFNITSSTCEL
SKQLNIAYDVTYSLACVRCCVNPFLYAFIGVKFRNDLFKLFKDLGCLSQEQLRQWSSCRH
IRRSSMSVEAETTTTFSP
|
|
|
|---|
| BDBM50088301 |
|---|
| n/a |
|---|
| Name | BDBM50088301 |
|---|
| Synonyms: | (E)-N,N-dimethyl-N-(4-(2-p-tolyl-6,7-dihydro-5H-benzo[7]annulene-8-carboxamido)benzyl)tetrahydro-2H-pyran-4-aminium chloride | (TAK779)Dimethyl-(tetrahydro-pyran-4-yl)-{4-[(3-p-tolyl-8,9-dihydro-7H-benzocycloheptene-6-carbonyl)-amino]-benzyl}-ammonium; chloride | CHEMBL41275 | CHEMBL64322 | Dimethyl-(tetrahydro-pyran-4-yl)-{4-[(3-p-tolyl-8,9-dihydro-7H-benzocycloheptene-6-carbonyl)-amino]-benzyl}-ammonium; chloride | Dimethyl-(tetrahydro-pyran-4-yl)-{4-[(3-p-tolyl-8,9-dihydro-7H-benzocycloheptene-6-carbonyl)-amino]-benzyl}-ammonium; chloride(TAK-779) | N-(4-(2-p-tolyl-6,7-dihydro-5H-benzo[7]annulene-8-carboxamido)benzyl)-N,N-dimethyl-tetrahydro-2H-pyran-4-aminium chloride | TAK-779 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H39N2O2 |
|---|
| Mol. Mass. | 495.6744 |
|---|
| SMILES | Cc1ccc(cc1)-c1ccc2CCCC(=Cc2c1)C(=O)Nc1ccc(C[N+](C)(C)C2CCOCC2)cc1 |c:15| |
|---|
| Structure | 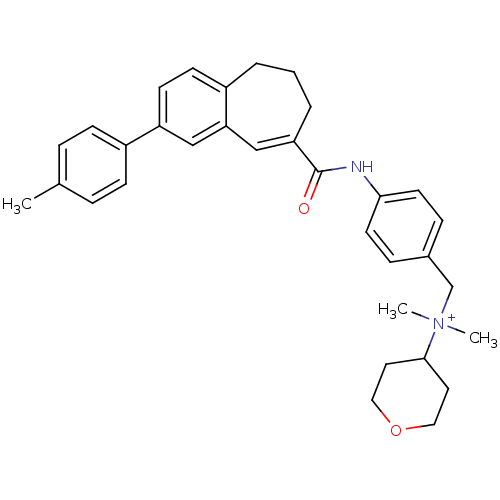
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Romero, FA; Jones, CT; Xu, Y; Fenaux, M; Halcomb, RL The Race to Bash NASH: Emerging Targets and Drug Development in a Complex Liver Disease. J Med Chem63:5031-5073 (2020) [PubMed] Article
Romero, FA; Jones, CT; Xu, Y; Fenaux, M; Halcomb, RL The Race to Bash NASH: Emerging Targets and Drug Development in a Complex Liver Disease. J Med Chem63:5031-5073 (2020) [PubMed] Article