| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin G/H synthase 1 |
|---|
| Ligand | BDBM50066569 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_158915 (CHEMBL763480) |
|---|
| IC50 | 35±n/a nM |
|---|
| Citation |  Janusz, JM; Young, PA; Ridgeway, JM; Scherz, MW; Enzweiler, K; Wu, LI; Gan, L; Chen, J; Kellstein, DE; Green, SA; Tulich, JL; Rosario-Jansen, T; Magrisso, IJ; Wehmeyer, KR; Kuhlenbeck, DL; Eichhold, TH; Dobson, RL New cyclooxygenase-2/5-lipoxygenase inhibitors. 3. 7-tert-butyl-2, 3-dihydro-3,3-dimethylbenzofuran derivatives as gastrointestinal safe antiinflammatory and analgesic agents: variations at the 5 position. J Med Chem41:3515-29 (1998) [PubMed] Article Janusz, JM; Young, PA; Ridgeway, JM; Scherz, MW; Enzweiler, K; Wu, LI; Gan, L; Chen, J; Kellstein, DE; Green, SA; Tulich, JL; Rosario-Jansen, T; Magrisso, IJ; Wehmeyer, KR; Kuhlenbeck, DL; Eichhold, TH; Dobson, RL New cyclooxygenase-2/5-lipoxygenase inhibitors. 3. 7-tert-butyl-2, 3-dihydro-3,3-dimethylbenzofuran derivatives as gastrointestinal safe antiinflammatory and analgesic agents: variations at the 5 position. J Med Chem41:3515-29 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin G/H synthase 1 |
|---|
| Name: | Prostaglandin G/H synthase 1 |
|---|
| Synonyms: | COX-1 | COX1 | Cyclooxygenase | Cyclooxygenase-1 | Cyclooxygenase-1 (COX-1) | PGH1_HUMAN | PTGS1 | Prostaglandin G/H synthase (cyclooxygenase) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 68692.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P23219 |
|---|
| Residue: | 599 |
|---|
| Sequence: | MSRSLLLWFLLFLLLLPPLPVLLADPGAPTPVNPCCYYPCQHQGICVRFGLDRYQCDCTR
TGYSGPNCTIPGLWTWLRNSLRPSPSFTHFLLTHGRWFWEFVNATFIREMLMRLVLTVRS
NLIPSPPTYNSAHDYISWESFSNVSYYTRILPSVPKDCPTPMGTKGKKQLPDAQLLARRF
LLRRKFIPDPQGTNLMFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDNLERQ
YQLRLFKDGKLKYQVLDGEMYPPSVEEAPVLMHYPRGIPPQSQMAVGQEVFGLLPGLMLY
ATLWLREHNRVCDLLKAEHPTWGDEQLFQTTRLILIGETIKIVIEEYVQQLSGYFLQLKF
DPELLFGVQFQYRNRIAMEFNHLYHWHPLMPDSFKVGSQEYSYEQFLFNTSMLVDYGVEA
LVDAFSRQIAGRIGGGRNMDHHILHVAVDVIRESREMRLQPFNEYRKRFGMKPYTSFQEL
VGEKEMAAELEELYGDIDALEFYPGLLLEKCHPNSIFGESMIEIGAPFSLKGLLGNPICS
PEYWKPSTFGGEVGFNIVKTATLKKLVCLNTKTCPYVSFRVPDASQDDGPAVERPSTEL
|
|
|
|---|
| BDBM50066569 |
|---|
| n/a |
|---|
| Name | BDBM50066569 |
|---|
| Synonyms: | (7-tert-Butyl-3,3-dimethyl-2,3-dihydro-benzofuran-5-yl)-(1-methyl-1H-pyrrol-2-yl)-methanone | CHEMBL119720 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H25NO2 |
|---|
| Mol. Mass. | 311.418 |
|---|
| SMILES | Cn1cccc1C(=O)c1cc2c(OCC2(C)C)c(c1)C(C)(C)C |
|---|
| Structure | 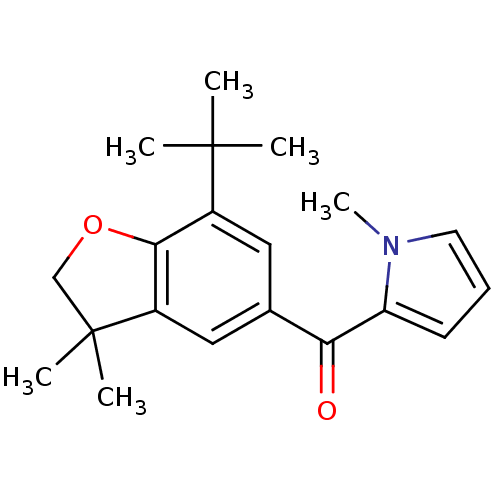
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Janusz, JM; Young, PA; Ridgeway, JM; Scherz, MW; Enzweiler, K; Wu, LI; Gan, L; Chen, J; Kellstein, DE; Green, SA; Tulich, JL; Rosario-Jansen, T; Magrisso, IJ; Wehmeyer, KR; Kuhlenbeck, DL; Eichhold, TH; Dobson, RL New cyclooxygenase-2/5-lipoxygenase inhibitors. 3. 7-tert-butyl-2, 3-dihydro-3,3-dimethylbenzofuran derivatives as gastrointestinal safe antiinflammatory and analgesic agents: variations at the 5 position. J Med Chem41:3515-29 (1998) [PubMed] Article
Janusz, JM; Young, PA; Ridgeway, JM; Scherz, MW; Enzweiler, K; Wu, LI; Gan, L; Chen, J; Kellstein, DE; Green, SA; Tulich, JL; Rosario-Jansen, T; Magrisso, IJ; Wehmeyer, KR; Kuhlenbeck, DL; Eichhold, TH; Dobson, RL New cyclooxygenase-2/5-lipoxygenase inhibitors. 3. 7-tert-butyl-2, 3-dihydro-3,3-dimethylbenzofuran derivatives as gastrointestinal safe antiinflammatory and analgesic agents: variations at the 5 position. J Med Chem41:3515-29 (1998) [PubMed] Article