| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Polyamine deacetylase HDAC10 |
|---|
| Ligand | BDBM50105327 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1875454 (CHEMBL4376743) |
|---|
| IC50 | 2.0±n/a nM |
|---|
| Citation |  Shouksmith, AE; Gawel, JM; Nawar, N; Sina, D; Raouf, YS; Bukhari, S; He, L; Johns, AE; Manaswiyoungkul, P; Olaoye, OO; Cabral, AD; Sedighi, A; de Araujo, ED; Gunning, PT Class I/IIb-Selective HDAC Inhibitor Exhibits Oral Bioavailability and Therapeutic Efficacy in Acute Myeloid Leukemia. ACS Med Chem Lett11:56-64 (2020) [PubMed] Article Shouksmith, AE; Gawel, JM; Nawar, N; Sina, D; Raouf, YS; Bukhari, S; He, L; Johns, AE; Manaswiyoungkul, P; Olaoye, OO; Cabral, AD; Sedighi, A; de Araujo, ED; Gunning, PT Class I/IIb-Selective HDAC Inhibitor Exhibits Oral Bioavailability and Therapeutic Efficacy in Acute Myeloid Leukemia. ACS Med Chem Lett11:56-64 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Polyamine deacetylase HDAC10 |
|---|
| Name: | Polyamine deacetylase HDAC10 |
|---|
| Synonyms: | HD10 | HDA10_HUMAN | HDAC10 | Histone deacetylase | Histone deacetylase 10 | Human HDAC10 |
|---|
| Type: | Chromatin regulator; hydrolase; repressor |
|---|
| Mol. Mass.: | 71431.89 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q969S8 |
|---|
| Residue: | 669 |
|---|
| Sequence: | MGTALVYHEDMTATRLLWDDPECEIERPERLTAALDRLRQRGLEQRCLRLSAREASEEEL
GLVHSPEYVSLVRETQVLGKEELQALSGQFDAIYFHPSTFHCARLAAGAGLQLVDAVLTG
AVQNGLALVRPPGHHGQRAAANGFCVFNNVAIAAAHAKQKHGLHRILVVDWDVHHGQGIQ
YLFEDDPSVLYFSWHRYEHGRFWPFLRESDADAVGRGQGLGFTVNLPWNQVGMGNADYVA
AFLHLLLPLAFEFDPELVLVSAGFDSAIGDPEGQMQATPECFAHLTQLLQVLAGGRVCAV
LEGGYHLESLAESVCMTVQTLLGDPAPPLSGPMAPCQSALESIQSARAAQAPHWKSLQQQ
DVTAVPMSPSSHSPEGRPPPLLPGGPVCKAAASAPSSLLDQPCLCPAPSVRTAVALTTPD
ITLVLPPDVIQQEASALREETEAWARPHESLAREEALTALGKLLYLLDGMLDGQVNSGIA
ATPASAAAATLDVAVRRGLSHGAQRLLCVALGQLDRPPDLAHDGRSLWLNIRGKEAAALS
MFHVSTPLPVMTGGFLSCILGLVLPLAYGFQPDLVLVALGPGHGLQGPHAALLAAMLRGL
AGGRVLALLEENSTPQLAGILARVLNGEAPPSLGPSSVASPEDVQALMYLRGQLEPQWKM
LQCHPHLVA
|
|
|
|---|
| BDBM50105327 |
|---|
| n/a |
|---|
| Name | BDBM50105327 |
|---|
| Synonyms: | JNJ-26481585 | Quisinostat |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H26N6O2 |
|---|
| Mol. Mass. | 394.4701 |
|---|
| SMILES | Cn1cc(CNCC2CCN(CC2)c2ncc(cn2)C(=O)NO)c2ccccc12 |
|---|
| Structure | 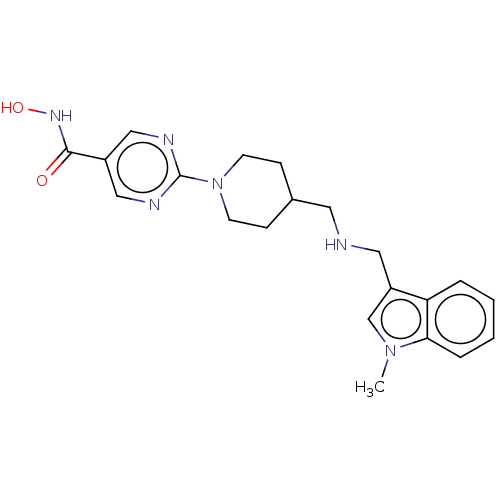
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Shouksmith, AE; Gawel, JM; Nawar, N; Sina, D; Raouf, YS; Bukhari, S; He, L; Johns, AE; Manaswiyoungkul, P; Olaoye, OO; Cabral, AD; Sedighi, A; de Araujo, ED; Gunning, PT Class I/IIb-Selective HDAC Inhibitor Exhibits Oral Bioavailability and Therapeutic Efficacy in Acute Myeloid Leukemia. ACS Med Chem Lett11:56-64 (2020) [PubMed] Article
Shouksmith, AE; Gawel, JM; Nawar, N; Sina, D; Raouf, YS; Bukhari, S; He, L; Johns, AE; Manaswiyoungkul, P; Olaoye, OO; Cabral, AD; Sedighi, A; de Araujo, ED; Gunning, PT Class I/IIb-Selective HDAC Inhibitor Exhibits Oral Bioavailability and Therapeutic Efficacy in Acute Myeloid Leukemia. ACS Med Chem Lett11:56-64 (2020) [PubMed] Article