| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1B |
|---|
| Ligand | BDBM50060426 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1616 (CHEMBL616641) |
|---|
| IC50 | 1.3±n/a nM |
|---|
| Citation |  Sternfeld, F; Guiblin, AR; Jelley, RA; Matassa, VG; Reeve, AJ; Hunt, PA; Beer, MS; Heald, A; Stanton, JA; Sohal, B; Watt, AP; Street, LJ Synthesis and serotonergic activity of 3-[2-(pyrrolidin-1-yl)ethyl]indoles: potent agonists for the h5-HT1D receptor with high selectivity over the h5-HT1B receptor. J Med Chem42:677-90 (1999) [PubMed] Article Sternfeld, F; Guiblin, AR; Jelley, RA; Matassa, VG; Reeve, AJ; Hunt, PA; Beer, MS; Heald, A; Stanton, JA; Sohal, B; Watt, AP; Street, LJ Synthesis and serotonergic activity of 3-[2-(pyrrolidin-1-yl)ethyl]indoles: potent agonists for the h5-HT1D receptor with high selectivity over the h5-HT1B receptor. J Med Chem42:677-90 (1999) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1B |
|---|
| Name: | 5-hydroxytryptamine receptor 1B |
|---|
| Synonyms: | 5-HT-1B | 5-HT-1D-beta | 5-HT1B | 5-hydroxytryptamine receptor 1B (5-HT1B) | 5HT1B_HUMAN | HTR1B | HTR1DB | S12 | Serotonin (5-HT) receptor | Serotonin 1D beta receptor | Serotonin Receptor 1B |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 43579.17 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Receptor binding assays were performed using human clone stably expressed in CHO cells |
|---|
| Residue: | 390 |
|---|
| Sequence: | MEEPGAQCAPPPPAGSETWVPQANLSSAPSQNCSAKDYIYQDSISLPWKVLLVMLLALIT
LATTLSNAFVIATVYRTRKLHTPANYLIASLAVTDLLVSILVMPISTMYTVTGRWTLGQV
VCDFWLSSDITCCTASILHLCVIALDRYWAITDAVEYSAKRTPKRAAVMIALVWVFSISI
SLPPFFWRQAKAEEEVSECVVNTDHILYTVYSTVGAFYFPTLLLIALYGRIYVEARSRIL
KQTPNRTGKRLTRAQLITDSPGSTSSVTSINSRVPDVPSESGSPVYVNQVKVRVSDALLE
KKKLMAARERKATKTLGIILGAFIVCWLPFFIISLVMPICKDACWFHLAIFDFFTWLGYL
NSLINPIIYTMSNEDFKQAFHKLIRFKCTS
|
|
|
|---|
| BDBM50060426 |
|---|
| n/a |
|---|
| Name | BDBM50060426 |
|---|
| Synonyms: | CHEMBL296161 | Dimethyl-[2-(5-[1,2,4]triazol-4-yl-1H-indol-3-yl)-ethyl]-amine | L-741604 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H17N5 |
|---|
| Mol. Mass. | 255.3183 |
|---|
| SMILES | CN(C)CCc1c[nH]c2ccc(cc12)-n1cnnc1 |
|---|
| Structure | 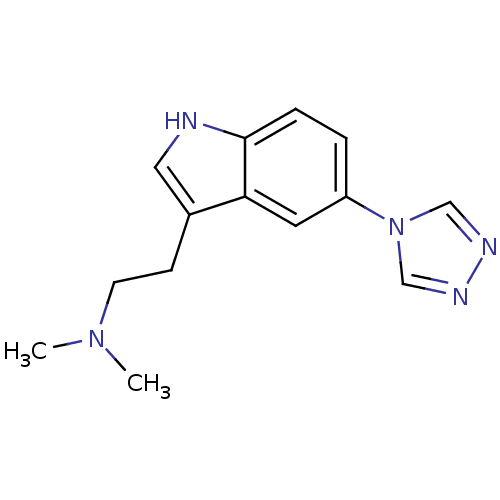
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sternfeld, F; Guiblin, AR; Jelley, RA; Matassa, VG; Reeve, AJ; Hunt, PA; Beer, MS; Heald, A; Stanton, JA; Sohal, B; Watt, AP; Street, LJ Synthesis and serotonergic activity of 3-[2-(pyrrolidin-1-yl)ethyl]indoles: potent agonists for the h5-HT1D receptor with high selectivity over the h5-HT1B receptor. J Med Chem42:677-90 (1999) [PubMed] Article
Sternfeld, F; Guiblin, AR; Jelley, RA; Matassa, VG; Reeve, AJ; Hunt, PA; Beer, MS; Heald, A; Stanton, JA; Sohal, B; Watt, AP; Street, LJ Synthesis and serotonergic activity of 3-[2-(pyrrolidin-1-yl)ethyl]indoles: potent agonists for the h5-HT1D receptor with high selectivity over the h5-HT1B receptor. J Med Chem42:677-90 (1999) [PubMed] Article