| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Somatostatin receptor type 2 |
|---|
| Ligand | BDBM50075289 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_200690 |
|---|
| Ki | 10±n/a nM |
|---|
| Citation |  Pasternak, A; Pan, Y; Marino, D; Sanderson, PE; Mosley, R; Rohrer, SP; Birzin, ET; Huskey, SE; Jacks, T; Schleim, KD; Cheng, K; Schaeffer, JM; Patchett, AA; Yang, L Potent, orally bioavailable somatostatin agonists: good absorption achieved by urea backbone cyclization. Bioorg Med Chem Lett9:491-6 (1999) [PubMed] Pasternak, A; Pan, Y; Marino, D; Sanderson, PE; Mosley, R; Rohrer, SP; Birzin, ET; Huskey, SE; Jacks, T; Schleim, KD; Cheng, K; Schaeffer, JM; Patchett, AA; Yang, L Potent, orally bioavailable somatostatin agonists: good absorption achieved by urea backbone cyclization. Bioorg Med Chem Lett9:491-6 (1999) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Somatostatin receptor type 2 |
|---|
| Name: | Somatostatin receptor type 2 |
|---|
| Synonyms: | SOMATOSTATIN SST2 | SSR2_MOUSE | Smstr2 | Somatostatin receptor | Somatostatin receptor 2 | Somatostatin receptor type 2 | Sst2 | Sstr2 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 41234.26 |
|---|
| Organism: | MOUSE |
|---|
| Description: | SOMATOSTATIN SST2 SSTR2 MOUSE::P30875 |
|---|
| Residue: | 369 |
|---|
| Sequence: | MEMSSEQLNGSQVWVSSPFDLNGSLGPSNGSNQTEPYYDMTSNAVLTFIYFVVCVVGLCG
NTLVIYVILRYAKMKTITNIYILNLAIADELFMLGLPFLAMQVALVHWPFGKAICRVVMT
VDGINQFTSIFCLTVMSIDRYLAVVHPIKSAKWRRPRTAKMINVAVWCVSLLVILPIMIY
AGLRSNQWGRSSCTINWPGESGAWYTGFIIYAFILGFLVPLTIICLCYLFIIIKVKSSGI
RVGSSKRKKSEKKVTRMVSIVVAVFIFCWLPFYIFNVSSVSVAISPTPALKGMFDFVVIL
TYANSCANPILYAFLSDNFKKSFQNVLCLVKVSGTEDGERSDSKQDKSRLNETTETQRTL
LNGDLQTSI
|
|
|
|---|
| BDBM50075289 |
|---|
| n/a |
|---|
| Name | BDBM50075289 |
|---|
| Synonyms: | 1-[5-amino-1-methyloxycarbonyl-(1R)-pentylcarbamoyl]-2-(1H-3-indolyl)-(1R)-ethyl spiro[2,3-dihydro-1H-indene-1,4'-(hexahydropyridine)]-1'-carboxylate | CHEMBL146022 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H40N4O5 |
|---|
| Mol. Mass. | 560.6838 |
|---|
| SMILES | COC(=O)[C@@H](CCCCN)NC(=O)[C@@H](Cc1c[nH]c2ccccc12)OC(=O)N1CCC2(CCc3ccccc23)CC1 |
|---|
| Structure | 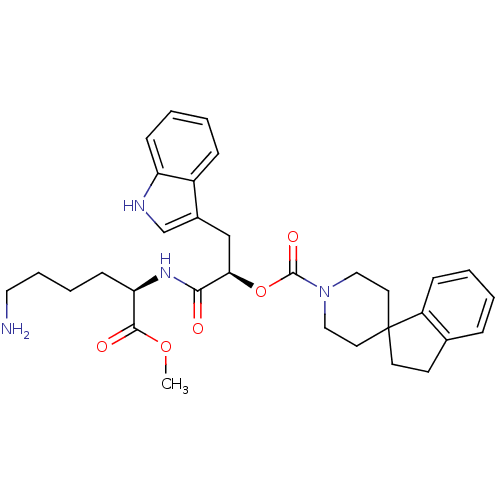
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pasternak, A; Pan, Y; Marino, D; Sanderson, PE; Mosley, R; Rohrer, SP; Birzin, ET; Huskey, SE; Jacks, T; Schleim, KD; Cheng, K; Schaeffer, JM; Patchett, AA; Yang, L Potent, orally bioavailable somatostatin agonists: good absorption achieved by urea backbone cyclization. Bioorg Med Chem Lett9:491-6 (1999) [PubMed]
Pasternak, A; Pan, Y; Marino, D; Sanderson, PE; Mosley, R; Rohrer, SP; Birzin, ET; Huskey, SE; Jacks, T; Schleim, KD; Cheng, K; Schaeffer, JM; Patchett, AA; Yang, L Potent, orally bioavailable somatostatin agonists: good absorption achieved by urea backbone cyclization. Bioorg Med Chem Lett9:491-6 (1999) [PubMed]