| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Carbonic anhydrase 5B, mitochondrial |
|---|
| Ligand | BDBM50528169 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1904505 (CHEMBL4406727) |
|---|
| Ki | 15±n/a nM |
|---|
| Citation |  Nocentini, A; Alterio, V; Bua, S; Micheli, L; Esposito, D; Buonanno, M; Bartolucci, G; Osman, SM; ALOthman, ZA; Cirilli, R; Pierini, M; Monti, SM; Di Cesare Mannelli, L; Gratteri, P; Ghelardini, C; De Simone, G; Supuran, CT Phenyl(thio)phosphon(amid)ate Benzenesulfonamides as Potent and Selective Inhibitors of Human Carbonic Anhydrases II and VII Counteract Allodynia in a Mouse Model of Oxaliplatin-Induced Neuropathy. J Med Chem63:5185-5200 (2020) [PubMed] Article Nocentini, A; Alterio, V; Bua, S; Micheli, L; Esposito, D; Buonanno, M; Bartolucci, G; Osman, SM; ALOthman, ZA; Cirilli, R; Pierini, M; Monti, SM; Di Cesare Mannelli, L; Gratteri, P; Ghelardini, C; De Simone, G; Supuran, CT Phenyl(thio)phosphon(amid)ate Benzenesulfonamides as Potent and Selective Inhibitors of Human Carbonic Anhydrases II and VII Counteract Allodynia in a Mouse Model of Oxaliplatin-Induced Neuropathy. J Med Chem63:5185-5200 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Carbonic anhydrase 5B, mitochondrial |
|---|
| Name: | Carbonic anhydrase 5B, mitochondrial |
|---|
| Synonyms: | CA-VB | CA5B | CAH5B_HUMAN | Carbonate dehydratase VB | Carbonic Anhydrase VB | Carbonic anhydrase 5B (CA VB) | Carbonic anhydrase 5B, mitochondrial | Carbonic anhydrase 5B, mitochondrial precursor | Carbonic anhydrase V | Carbonic anhydrase VB (CA VB) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 36440.83 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human (cloned) isozyme |
|---|
| Residue: | 317 |
|---|
| Sequence: | MVVMNSLRVILQASPGKLLWRKFQIPRFMPARPCSLYTCTYKTRNRALHPLWESVDLVPG
GDRQSPINIRWRDSVYDPGLKPLTISYDPATCLHVWNNGYSFLVEFEDSTDKSVIKGGPL
EHNYRLKQFHFHWGAIDAWGSEHTVDSKCFPAELHLVHWNAVRFENFEDAALEENGLAVI
GVFLKLGKHHKELQKLVDTLPSIKHKDALVEFGSFDPSCLMPTCPDYWTYSGSLTTPPLS
ESVTWIIKKQPVEVDHDQLEQFRTLLFTSEGEKEKRMVDNFRPLQPLMNRTVRSSFRHDY
VLNVQAKPKPATSQATP
|
|
|
|---|
| BDBM50528169 |
|---|
| n/a |
|---|
| Name | BDBM50528169 |
|---|
| Synonyms: | CHEMBL4579472 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H17N2O7PS2 |
|---|
| Mol. Mass. | 468.441 |
|---|
| SMILES | NS(=O)(=O)c1ccc(OP(=O)(Oc2ccc(cc2)S(N)(=O)=O)c2ccccc2)cc1 |
|---|
| Structure | 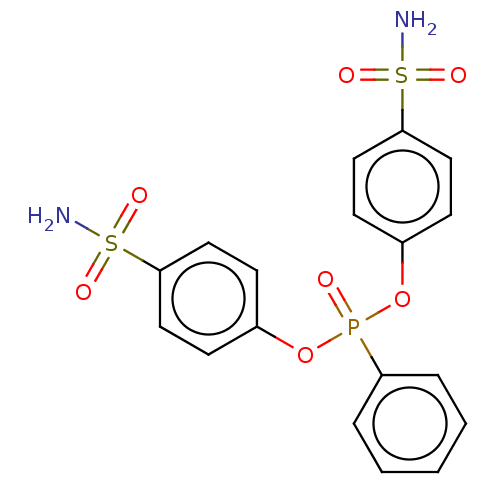
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nocentini, A; Alterio, V; Bua, S; Micheli, L; Esposito, D; Buonanno, M; Bartolucci, G; Osman, SM; ALOthman, ZA; Cirilli, R; Pierini, M; Monti, SM; Di Cesare Mannelli, L; Gratteri, P; Ghelardini, C; De Simone, G; Supuran, CT Phenyl(thio)phosphon(amid)ate Benzenesulfonamides as Potent and Selective Inhibitors of Human Carbonic Anhydrases II and VII Counteract Allodynia in a Mouse Model of Oxaliplatin-Induced Neuropathy. J Med Chem63:5185-5200 (2020) [PubMed] Article
Nocentini, A; Alterio, V; Bua, S; Micheli, L; Esposito, D; Buonanno, M; Bartolucci, G; Osman, SM; ALOthman, ZA; Cirilli, R; Pierini, M; Monti, SM; Di Cesare Mannelli, L; Gratteri, P; Ghelardini, C; De Simone, G; Supuran, CT Phenyl(thio)phosphon(amid)ate Benzenesulfonamides as Potent and Selective Inhibitors of Human Carbonic Anhydrases II and VII Counteract Allodynia in a Mouse Model of Oxaliplatin-Induced Neuropathy. J Med Chem63:5185-5200 (2020) [PubMed] Article