| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mu-type opioid receptor |
|---|
| Ligand | BDBM50008984 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1908619 (CHEMBL4410977) |
|---|
| EC50 | 0.510000±n/a nM |
|---|
| Citation |  Hsu, FL; Walz, AJ; Myslinski, JM; Kong, L; Feasel, MG; Goralski, TDP; Rose, T; Cooper, NJ; Roughley, N; Timperley, CM Synthesis and ?-Opioid Activity of the Primary Metabolites of Carfentanil. ACS Med Chem Lett10:1568-1572 (2019) [PubMed] Article Hsu, FL; Walz, AJ; Myslinski, JM; Kong, L; Feasel, MG; Goralski, TDP; Rose, T; Cooper, NJ; Roughley, N; Timperley, CM Synthesis and ?-Opioid Activity of the Primary Metabolites of Carfentanil. ACS Med Chem Lett10:1568-1572 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mu-type opioid receptor |
|---|
| Name: | Mu-type opioid receptor |
|---|
| Synonyms: | M-OR-1 | MOP | MOR-1 | MOR1 | MUOR1 | Mu Opioid Receptor | Mu opiate receptor | OPIATE Mu | OPRM1 | OPRM_HUMAN | hMOP | mu-type opioid receptor isoform MOR-1 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44789.51 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P35372 |
|---|
| Residue: | 400 |
|---|
| Sequence: | MDSSAAPTNASNCTDALAYSSCSPAPSPGSWVNLSHLDGNLSDPCGPNRTDLGGRDSLCP
PTGSPSMITAITIMALYSIVCVVGLFGNFLVMYVIVRYTKMKTATNIYIFNLALADALAT
STLPFQSVNYLMGTWPFGTILCKIVISIDYYNMFTSIFTLCTMSVDRYIAVCHPVKALDF
RTPRNAKIINVCNWILSSAIGLPVMFMATTKYRQGSIDCTLTFSHPTWYWENLLKICVFI
FAFIMPVLIITVCYGLMILRLKSVRMLSGSKEKDRNLRRITRMVLVVVAVFIVCWTPIHI
YVIIKALVTIPETTFQTVSWHFCIALGYTNSCLNPVLYAFLDENFKRCFREFCIPTSSNI
EQQNSTRIRQNTRDHPSTANTVDRTNHQLENLEAETAPLP
|
|
|
|---|
| BDBM50008984 |
|---|
| n/a |
|---|
| Name | BDBM50008984 |
|---|
| Synonyms: | 4-(4-Chloro-benzyl)-2-(1-methyl-azepan-4-yl)-2H-phthalazin-1-one | CHEMBL596 | Duragesic-100 | Duragesic-12 | Duragesic-25 | Duragesic-50 | Duragesic-75 | FENTANYL | FENTANYL CITRATE | FENTANYL-HCl | Fentanyl-100 | Fentanyl-12 | Fentanyl-25 | Fentanyl-50 | Fentanyl-75 | Fentora | Innovar | Ionsys | N-(1-Phenethyl-piperidin-4-yl)-N-phenyl-propionamide | N-(1-Phenethyl-piperidin-4-yl)-N-phenyl-propionamide(Fentanyl) | N-(1-phenethylpiperidin-4-yl)-N-phenylpropionamide | US20230399418, Compound Fentanyl |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H28N2O |
|---|
| Mol. Mass. | 336.4705 |
|---|
| SMILES | CCC(=O)N(C1CCN(CCc2ccccc2)CC1)c1ccccc1 |
|---|
| Structure | 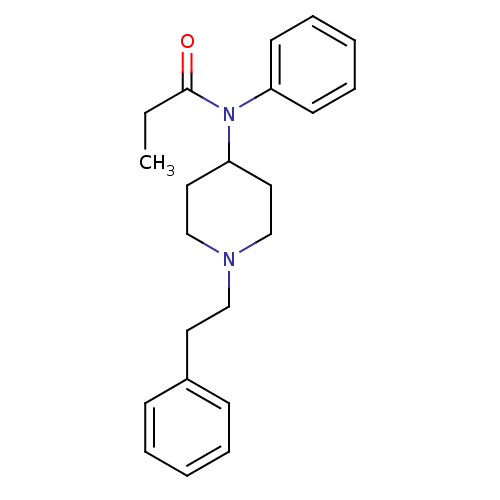
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hsu, FL; Walz, AJ; Myslinski, JM; Kong, L; Feasel, MG; Goralski, TDP; Rose, T; Cooper, NJ; Roughley, N; Timperley, CM Synthesis and ?-Opioid Activity of the Primary Metabolites of Carfentanil. ACS Med Chem Lett10:1568-1572 (2019) [PubMed] Article
Hsu, FL; Walz, AJ; Myslinski, JM; Kong, L; Feasel, MG; Goralski, TDP; Rose, T; Cooper, NJ; Roughley, N; Timperley, CM Synthesis and ?-Opioid Activity of the Primary Metabolites of Carfentanil. ACS Med Chem Lett10:1568-1572 (2019) [PubMed] Article