

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Neuropeptide Y receptor type 5 | ||
| Ligand | BDBM50089069 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEBML_143988 | ||
| IC50 | 2±n/a nM | ||
| Citation |  Rueeger, H; Rigollier, P; Yamaguchi, Y; Schmidlin, T; Schilling, W; Criscione, L; Whitebread, S; Chiesi, M; Walker, MW; Dhanoa, D; Islam, I; Zhang, J; Gluchowski, C Design, synthesis and SAR of a series of 2-substituted 4-amino-quinazoline neuropeptide Y Y5 receptor antagonists. Bioorg Med Chem Lett10:1175-9 (2000) [PubMed] Rueeger, H; Rigollier, P; Yamaguchi, Y; Schmidlin, T; Schilling, W; Criscione, L; Whitebread, S; Chiesi, M; Walker, MW; Dhanoa, D; Islam, I; Zhang, J; Gluchowski, C Design, synthesis and SAR of a series of 2-substituted 4-amino-quinazoline neuropeptide Y Y5 receptor antagonists. Bioorg Med Chem Lett10:1175-9 (2000) [PubMed] | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Neuropeptide Y receptor type 5 | |||
| Name: | Neuropeptide Y receptor type 5 | ||
| Synonyms: | NPY-Y5 | NPY-Y5 receptor | NPY5-R | NPY5R | NPY5R_HUMAN | NPYR5 | NPYY5 | Neuropeptide Y receptor type 5 | Neuropeptide Y receptor type 5 ( NPY Y5) | Y5 receptor | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 50746.64 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | Q15761 | ||
| Residue: | 445 | ||
| Sequence: |
| ||
| BDBM50089069 | |||
| n/a | |||
| Name | BDBM50089069 | ||
| Synonyms: | CHEMBL17632 | N-[4-(4-Phenylamino-quinazolin-2-ylamino)-cyclohexylmethyl]-methanesulfonamide | ||
| Type | Small organic molecule | ||
| Emp. Form. | C22H27N5O2S | ||
| Mol. Mass. | 425.547 | ||
| SMILES | CS(=O)(=O)NC[C@H]1CC[C@@H](CC1)Nc1nc(Nc2ccccc2)c2ccccc2n1 |wU:9.12,wD:6.5,(17,-10.36,;16.58,-8.88,;18,-9.04,;15.26,-9.64,;16.58,-7.34,;15.26,-6.57,;13.94,-7.34,;12.59,-6.57,;11.24,-7.34,;11.27,-8.89,;12.61,-9.64,;13.94,-8.88,;9.94,-9.67,;8.6,-8.89,;8.6,-7.34,;7.25,-6.58,;7.25,-5.05,;8.57,-4.26,;8.55,-2.74,;9.87,-1.96,;11.22,-2.72,;11.24,-4.26,;9.9,-5.04,;5.93,-7.36,;4.6,-6.59,;3.26,-7.36,;3.26,-8.9,;4.6,-9.68,;5.93,-8.9,;7.27,-9.67,)| | ||
| Structure | 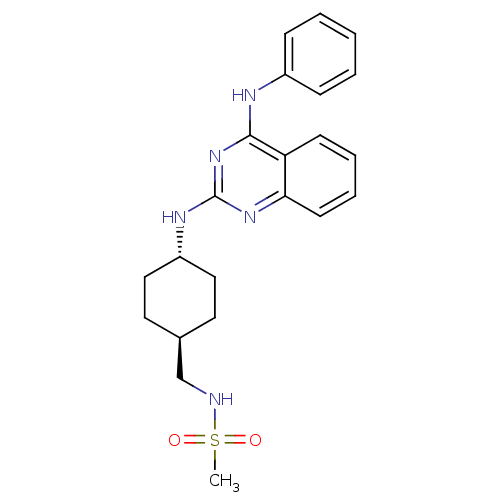
| ||