| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuropeptide Y receptor type 5 |
|---|
| Ligand | BDBM50101563 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_143298 (CHEMBL872841) |
|---|
| IC50 | 6.5±n/a nM |
|---|
| Citation |  Fotsch, C; Sonnenberg, JD; Chen, N; Hale, C; Karbon, W; Norman, MH Synthesis and structure-activity relationships of trisubstituted phenyl urea derivatives as neuropeptide Y5 receptor antagonists. J Med Chem44:2344-56 (2001) [PubMed] Fotsch, C; Sonnenberg, JD; Chen, N; Hale, C; Karbon, W; Norman, MH Synthesis and structure-activity relationships of trisubstituted phenyl urea derivatives as neuropeptide Y5 receptor antagonists. J Med Chem44:2344-56 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuropeptide Y receptor type 5 |
|---|
| Name: | Neuropeptide Y receptor type 5 |
|---|
| Synonyms: | NPY-Y5 | NPY-Y5 receptor | NPY5-R | NPY5R | NPY5R_HUMAN | NPYR5 | NPYY5 | Neuropeptide Y receptor type 5 | Neuropeptide Y receptor type 5 ( NPY Y5) | Y5 receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 50746.64 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q15761 |
|---|
| Residue: | 445 |
|---|
| Sequence: | MDLELDEYYNKTLATENNTAATRNSDFPVWDDYKSSVDDLQYFLIGLYTFVSLLGFMGNL
LILMALMKKRNQKTTVNFLIGNLAFSDILVVLFCSPFTLTSVLLDQWMFGKVMCHIMPFL
QCVSVLVSTLILISIAIVRYHMIKHPISNNLTANHGYFLIATVWTLGFAICSPLPVFHSL
VELQETFGSALLSSRYLCVESWPSDSYRIAFTISLLLVQYILPLVCLTVSHTSVCRSISC
GLSNKENRLEENEMINLTLHPSKKSGPQVKLSGSHKWSYSFIKKHRRRYSKKTACVLPAP
ERPSQENHSRILPENFGSVRSQLSSSSKFIPGVPTCFEIKPEENSDVHELRVKRSVTRIK
KRSRSVFYRLTILILVFAVSWMPLHLFHVVTDFNDNLISNRHFKLVYCICHLLGMMSCCL
NPILYGFLNNGIKADLVSLIHCLHM
|
|
|
|---|
| BDBM50101563 |
|---|
| n/a |
|---|
| Name | BDBM50101563 |
|---|
| Synonyms: | 1-(2-Hydroxy-1-methyl-2-phenyl-ethyl)-1-methyl-3-(4-phenoxy-phenyl)-urea | CHEMBL77639 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H24N2O3 |
|---|
| Mol. Mass. | 376.4483 |
|---|
| SMILES | C[C@H]([C@@H](O)c1ccccc1)N(C)C(=O)Nc1ccc(Oc2ccccc2)cc1 |
|---|
| Structure | 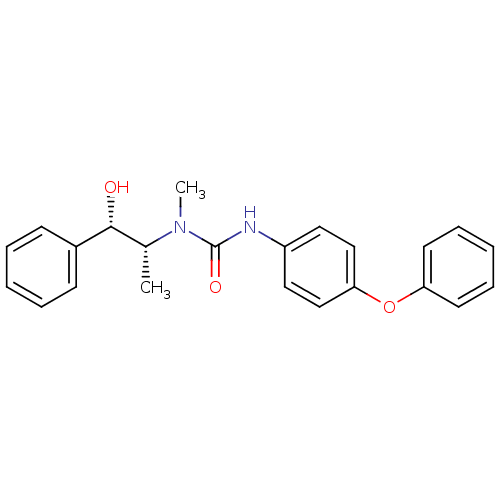
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fotsch, C; Sonnenberg, JD; Chen, N; Hale, C; Karbon, W; Norman, MH Synthesis and structure-activity relationships of trisubstituted phenyl urea derivatives as neuropeptide Y5 receptor antagonists. J Med Chem44:2344-56 (2001) [PubMed]
Fotsch, C; Sonnenberg, JD; Chen, N; Hale, C; Karbon, W; Norman, MH Synthesis and structure-activity relationships of trisubstituted phenyl urea derivatives as neuropeptide Y5 receptor antagonists. J Med Chem44:2344-56 (2001) [PubMed]