| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Myeloperoxidase |
|---|
| Ligand | BDBM50554050 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2048419 (CHEMBL4703118) |
|---|
| IC50 | 2.9±n/a nM |
|---|
| Citation |  Shaw, SA; Vokits, BP; Dilger, AK; Viet, A; Clark, CG; Abell, LM; Locke, GA; Duke, G; Kopcho, LM; Dongre, A; Gao, J; Krishnakumar, A; Jusuf, S; Khan, J; Spronk, SA; Basso, MD; Zhao, L; Cantor, GH; Onorato, JM; Wexler, RR; Duclos, F; Kick, EK Discovery and structure activity relationships of 7-benzyl triazolopyridines as stable, selective, and reversible inhibitors of myeloperoxidase. Bioorg Med Chem28:0 (2020) [PubMed] Article Shaw, SA; Vokits, BP; Dilger, AK; Viet, A; Clark, CG; Abell, LM; Locke, GA; Duke, G; Kopcho, LM; Dongre, A; Gao, J; Krishnakumar, A; Jusuf, S; Khan, J; Spronk, SA; Basso, MD; Zhao, L; Cantor, GH; Onorato, JM; Wexler, RR; Duclos, F; Kick, EK Discovery and structure activity relationships of 7-benzyl triazolopyridines as stable, selective, and reversible inhibitors of myeloperoxidase. Bioorg Med Chem28:0 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Myeloperoxidase |
|---|
| Name: | Myeloperoxidase |
|---|
| Synonyms: | 84 kDa myeloperoxidase | 89 kDa myeloperoxidase | MPO | Mpo protein | Myeloperoxidase | Myeloperoxidase (MPO) | Myeloperoxidase heavy chain | Myeloperoxidase light chain | PERM_HUMAN |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 83888.32 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05164 |
|---|
| Residue: | 745 |
|---|
| Sequence: | MGVPFFSSLRCMVDLGPCWAGGLTAEMKLLLALAGLLAILATPQPSEGAAPAVLGEVDTS
LVLSSMEEAKQLVDKAYKERRESIKQRLRSGSASPMELLSYFKQPVAATRTAVRAADYLH
VALDLLERKLRSLWRRPFNVTDVLTPAQLNVLSKSSGCAYQDVGVTCPEQDKYRTITGMC
NNRRSPTLGASNRAFVRWLPAEYEDGFSLPYGWTPGVKRNGFPVALARAVSNEIVRFPTD
QLTPDQERSLMFMQWGQLLDHDLDFTPEPAARASFVTGVNCETSCVQQPPCFPLKIPPND
PRIKNQADCIPFFRSCPACPGSNITIRNQINALTSFVDASMVYGSEEPLARNLRNMSNQL
GLLAVNQRFQDNGRALLPFDNLHDDPCLLTNRSARIPCFLAGDTRSSEMPELTSMHTLLL
REHNRLATELKSLNPRWDGERLYQEARKIVGAMVQIITYRDYLPLVLGPTAMRKYLPTYR
SYNDSVDPRIANVFTNAFRYGHTLIQPFMFRLDNRYQPMEPNPRVPLSRVFFASWRVVLE
GGIDPILRGLMATPAKLNRQNQIAVDEIRERLFEQVMRIGLDLPALNMQRSRDHGLPGYN
AWRRFCGLPQPETVGQLGTVLRNLKLARKLMEQYGTPNNIDIWMGGVSEPLKRKGRVGPL
LACIIGTQFRKLRDGDRFWWENEGVFSMQQRQALAQISLPRIICDNTGITTVSKNNIFMS
NSYPRDFVNCSTLPALNLASWREAS
|
|
|
|---|
| BDBM50554050 |
|---|
| n/a |
|---|
| Name | BDBM50554050 |
|---|
| Synonyms: | CHEMBL4752248 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H30N6 |
|---|
| Mol. Mass. | 474.5994 |
|---|
| SMILES | Nc1cc([C@H](CCN[C@@H]2CC[C@@H](c3ccccc3)c3ccccc23)c2ccccc2)c2nn[nH]c2n1 |r| |
|---|
| Structure | 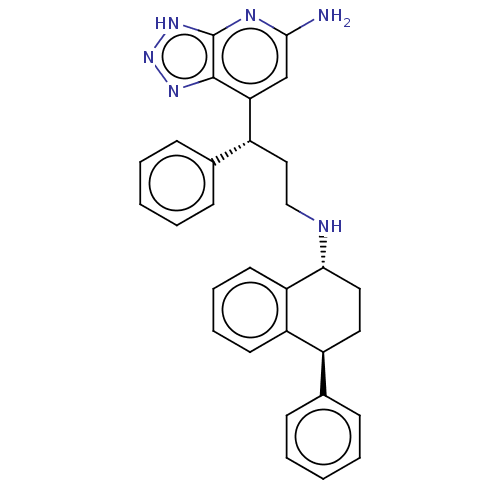
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Shaw, SA; Vokits, BP; Dilger, AK; Viet, A; Clark, CG; Abell, LM; Locke, GA; Duke, G; Kopcho, LM; Dongre, A; Gao, J; Krishnakumar, A; Jusuf, S; Khan, J; Spronk, SA; Basso, MD; Zhao, L; Cantor, GH; Onorato, JM; Wexler, RR; Duclos, F; Kick, EK Discovery and structure activity relationships of 7-benzyl triazolopyridines as stable, selective, and reversible inhibitors of myeloperoxidase. Bioorg Med Chem28:0 (2020) [PubMed] Article
Shaw, SA; Vokits, BP; Dilger, AK; Viet, A; Clark, CG; Abell, LM; Locke, GA; Duke, G; Kopcho, LM; Dongre, A; Gao, J; Krishnakumar, A; Jusuf, S; Khan, J; Spronk, SA; Basso, MD; Zhao, L; Cantor, GH; Onorato, JM; Wexler, RR; Duclos, F; Kick, EK Discovery and structure activity relationships of 7-benzyl triazolopyridines as stable, selective, and reversible inhibitors of myeloperoxidase. Bioorg Med Chem28:0 (2020) [PubMed] Article