| Reaction Details |
|---|
 | Report a problem with these data |
| Target | C-C chemokine receptor type 5 |
|---|
| Ligand | BDBM50123437 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_39648 (CHEMBL649953) |
|---|
| Ki | 11±n/a nM |
|---|
| Citation |  McCombie, SW; Tagat, JR; Vice, SF; Lin, SI; Steensma, R; Palani, A; Neustadt, BR; Baroudy, BM; Strizki, JM; Endres, M; Cox, K; Dan, N; Chou, CC Piperazine-based CCR5 antagonists as HIV-1 inhibitors. III: synthesis, antiviral and pharmacokinetic profiles of symmetrical heteroaryl carboxamides. Bioorg Med Chem Lett13:567-71 (2003) [PubMed] McCombie, SW; Tagat, JR; Vice, SF; Lin, SI; Steensma, R; Palani, A; Neustadt, BR; Baroudy, BM; Strizki, JM; Endres, M; Cox, K; Dan, N; Chou, CC Piperazine-based CCR5 antagonists as HIV-1 inhibitors. III: synthesis, antiviral and pharmacokinetic profiles of symmetrical heteroaryl carboxamides. Bioorg Med Chem Lett13:567-71 (2003) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| C-C chemokine receptor type 5 |
|---|
| Name: | C-C chemokine receptor type 5 |
|---|
| Synonyms: | C-C CKR-5 | C-C chemokine receptor type 5 | CC-CKR-5 | CCR-5 | CCR5 | CCR5/mu opioid receptor complex | CCR5_HUMAN | CD_antigen=CD195 | CHEMR13 | CMKBR5 | HIV-1 fusion coreceptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 40540.21 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P51681 |
|---|
| Residue: | 352 |
|---|
| Sequence: | MDYQVSSPIYDINYYTSEPCQKINVKQIAARLLPPLYSLVFIFGFVGNMLVILILINCKR
LKSMTDIYLLNLAISDLFFLLTVPFWAHYAAAQWDFGNTMCQLLTGLYFIGFFSGIFFII
LLTIDRYLAVVHAVFALKARTVTFGVVTSVITWVVAVFASLPGIIFTRSQKEGLHYTCSS
HFPYSQYQFWKNFQTLKIVILGLVLPLLVMVICYSGILKTLLRCRNEKKRHRAVRLIFTI
MIVYFLFWAPYNIVLLLNTFQEFFGLNNCSSSNRLDQAMQVTETLGMTHCCINPIIYAFV
GEKFRNYLLVFFQKHIAKRFCKCCSIFQQEAPERASSVYTRSTGEQEISVGL
|
|
|
|---|
| BDBM50123437 |
|---|
| n/a |
|---|
| Name | BDBM50123437 |
|---|
| Synonyms: | (2-Methoxy-4,6-dimethyl-pyrimidin-5-yl)-(4-methyl-4-{(S)-3-methyl-4-[(S)-1-(4-trifluoromethyl-phenyl)-ethyl]-piperazin-1-yl}-piperidin-1-yl)-methanone | CHEMBL140706 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H38F3N5O2 |
|---|
| Mol. Mass. | 533.6288 |
|---|
| SMILES | COc1nc(C)c(C(=O)N2CCC(C)(CC2)N2CCN([C@@H](C)c3ccc(cc3)C(F)(F)F)[C@@H](C)C2)c(C)n1 |
|---|
| Structure | 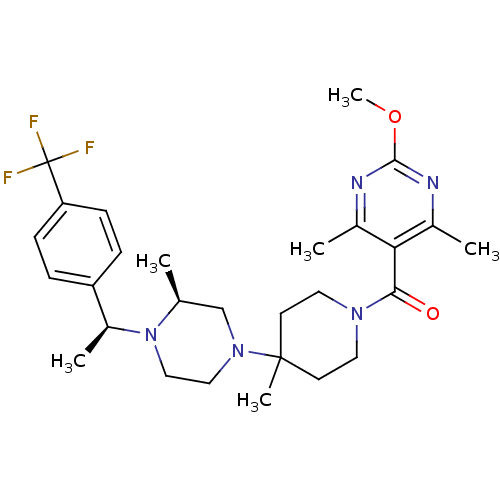
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 McCombie, SW; Tagat, JR; Vice, SF; Lin, SI; Steensma, R; Palani, A; Neustadt, BR; Baroudy, BM; Strizki, JM; Endres, M; Cox, K; Dan, N; Chou, CC Piperazine-based CCR5 antagonists as HIV-1 inhibitors. III: synthesis, antiviral and pharmacokinetic profiles of symmetrical heteroaryl carboxamides. Bioorg Med Chem Lett13:567-71 (2003) [PubMed]
McCombie, SW; Tagat, JR; Vice, SF; Lin, SI; Steensma, R; Palani, A; Neustadt, BR; Baroudy, BM; Strizki, JM; Endres, M; Cox, K; Dan, N; Chou, CC Piperazine-based CCR5 antagonists as HIV-1 inhibitors. III: synthesis, antiviral and pharmacokinetic profiles of symmetrical heteroaryl carboxamides. Bioorg Med Chem Lett13:567-71 (2003) [PubMed]