| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM50135159 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_27799 (CHEMBL636509) |
|---|
| Ki | 12200±n/a nM |
|---|
| Citation |  Zaheer-Ul-Haq, ZU; Wellenzohn, B; Liedl, KR; Rode, BM Molecular docking studies of natural cholinesterase-inhibiting steroidal alkaloids from Sarcococca saligna. J Med Chem46:5087-90 (2003) [PubMed] Article Zaheer-Ul-Haq, ZU; Wellenzohn, B; Liedl, KR; Rode, BM Molecular docking studies of natural cholinesterase-inhibiting steroidal alkaloids from Sarcococca saligna. J Med Chem46:5087-90 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | 3.1.1.7 | ACES_TETCF | Acetylcholinesterase (AChE) | Acetylcholinesterase precursor | ache |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 65900.74 |
|---|
| Organism: | Tetronarce californica (Pacific electric ray) (Torpedo californica) |
|---|
| Description: | P04058 |
|---|
| Residue: | 586 |
|---|
| Sequence: | MNLLVTSSLGVLLHLVVLCQADDHSELLVNTKSGKVMGTRVPVLSSHISAFLGIPFAEPP
VGNMRFRRPEPKKPWSGVWNASTYPNNCQQYVDEQFPGFSGSEMWNPNREMSEDCLYLNI
WVPSPRPKSTTVMVWIYGGGFYSGSSTLDVYNGKYLAYTEEVVLVSLSYRVGAFGFLALH
GSQEAPGNVGLLDQRMALQWVHDNIQFFGGDPKTVTIFGESAGGASVGMHILSPGSRDLF
RRAILQSGSPNCPWASVSVAEGRRRAVELGRNLNCNLNSDEELIHCLREKKPQELIDVEW
NVLPFDSIFRFSFVPVIDGEFFPTSLESMLNSGNFKKTQILLGVNKDEGSFFLLYGAPGF
SKDSESKISREDFMSGVKLSVPHANDLGLDAVTLQYTDWMDDNNGIKNRDGLDDIVGDHN
VICPLMHFVNKYTKFGNGTYLYFFNHRASNLVWPEWMGVIHGYEIEFVFGLPLVKELNYT
AEEEALSRRIMHYWATFAKTGNPNEPHSQESKWPLFTTKEQKFIDLNTEPMKVHQRLRVQ
MCVFWNQFLPKLLNATACDGELSSSGTSSSKGIIFYVLFSILYLIF
|
|
|
|---|
| BDBM50135159 |
|---|
| n/a |
|---|
| Name | BDBM50135159 |
|---|
| Synonyms: | (20S,2'E)-20-(N,N-dimethylamino)-3beta-(3'-phenyl-2'-propenyl-N-methylamido)pregnane] | (E)-N-[(3S,5S,8R,9S,10S,13S,14S,17S)-17-((S)-1-Dimethylamino-ethyl)-10,13-dimethyl-hexadecahydro-cyclopenta[a]phenanthren-3-yl]-N-methyl-3-phenyl-acrylamide | (E)-N-{(1S,6S,7S,11R,12S,15S,16S)-17-[(S)-1-((S)-Dimethyl-amino)-ethyl]-10,13-dimethyl-hexadecahydro-cyclopenta[a]phenanthren-3-yl}-N-methyl-3-phenyl-acrylamide | CHEMBL409854 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H50N2O |
|---|
| Mol. Mass. | 490.7629 |
|---|
| SMILES | C[C@@H]([C@H]1CC[C@H]2[C@@H]3CC[C@H]4C[C@H](CC[C@]4(C)[C@H]3CC[C@]12C)N(C)C(=O)\C=C\c1ccccc1)N(C)C |r| |
|---|
| Structure | 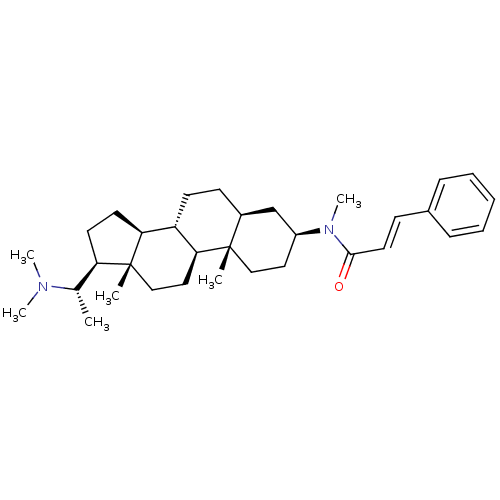
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zaheer-Ul-Haq, ZU; Wellenzohn, B; Liedl, KR; Rode, BM Molecular docking studies of natural cholinesterase-inhibiting steroidal alkaloids from Sarcococca saligna. J Med Chem46:5087-90 (2003) [PubMed] Article
Zaheer-Ul-Haq, ZU; Wellenzohn, B; Liedl, KR; Rode, BM Molecular docking studies of natural cholinesterase-inhibiting steroidal alkaloids from Sarcococca saligna. J Med Chem46:5087-90 (2003) [PubMed] Article