| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor VII |
|---|
| Ligand | BDBM50572517 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2117196 (CHEMBL4826262) |
|---|
| Ki | 4193±n/a nM |
|---|
| Citation |  Lei, Y; Zhang, B; Zhang, Y; Dai, X; Duan, Y; Mao, Q; Gao, J; Yang, Y; Bao, Z; Fu, X; Ping, K; Yan, C; Mou, Y; Wang, S Design, synthesis and biological evaluation of novel FXIa inhibitors with 2-phenyl-1H-imidazole-5-carboxamide moiety as P1 fragment. Eur J Med Chem220:0 (2021) [PubMed] Article Lei, Y; Zhang, B; Zhang, Y; Dai, X; Duan, Y; Mao, Q; Gao, J; Yang, Y; Bao, Z; Fu, X; Ping, K; Yan, C; Mou, Y; Wang, S Design, synthesis and biological evaluation of novel FXIa inhibitors with 2-phenyl-1H-imidazole-5-carboxamide moiety as P1 fragment. Eur J Med Chem220:0 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor VII |
|---|
| Name: | Coagulation factor VII |
|---|
| Synonyms: | Eptacog alfa | F7 | FA7_HUMAN | Factor VIIa | Factor VIIa (fVIIa) | Proconvertin | SPCA | Thrombin and coagulation factor VII | serum prothrombin conversion accelerator |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 51599.89 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 466 |
|---|
| Sequence: | MVSQALRLLCLLLGLQGCLAAGGVAKASGGETRDMPWKPGPHRVFVTQEEAHGVLHRRRR
ANAFLEELRPGSLERECKEEQCSFEEAREIFKDAERTKLFWISYSDGDQCASSPCQNGGS
CKDQLQSYICFCLPAFEGRNCETHKDDQLICVNENGGCEQYCSDHTGTKRSCRCHEGYSL
LADGVSCTPTVEYPCGKIPILEKRNASKPQGRIVGGKVCPKGECPWQVLLLVNGAQLCGG
TLINTIWVVSAAHCFDKIKNWRNLIAVLGEHDLSEHDGDEQSRRVAQVIIPSTYVPGTTN
HDIALLRLHQPVVLTDHVVPLCLPERTFSERTLAFVRFSLVSGWGQLLDRGATALELMVL
NVPRLMTQDCLQQSRKVGDSPNITEYMFCAGYSDGSKDSCKGDSGGPHATHYRGTWYLTG
IVSWGQGCATVGHFGVYTRVSQYIEWLQKLMRSEPRPGVLLRAPFP
|
|
|
|---|
| BDBM50572517 |
|---|
| n/a |
|---|
| Name | BDBM50572517 |
|---|
| Synonyms: | CHEMBL4865339 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C34H30ClFN6O6 |
|---|
| Mol. Mass. | 673.09 |
|---|
| SMILES | CC1CC1C(=O)Nc1ccc(C[C@H](NC(=O)c2c(C)nc(-c3cccc(Cl)c3F)n2O)C(=O)Nc2ccc3[nH]c(cc3c2)C(O)=O)cc1 |r| |
|---|
| Structure | 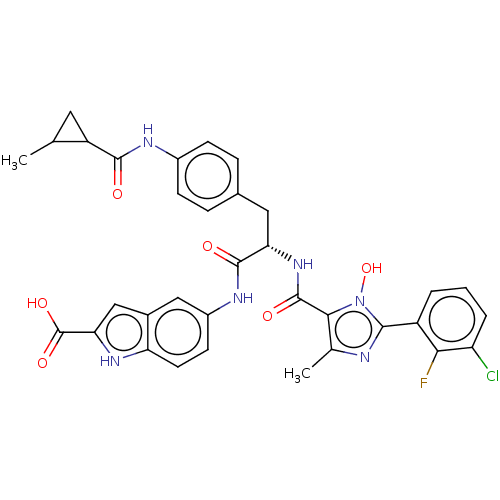
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lei, Y; Zhang, B; Zhang, Y; Dai, X; Duan, Y; Mao, Q; Gao, J; Yang, Y; Bao, Z; Fu, X; Ping, K; Yan, C; Mou, Y; Wang, S Design, synthesis and biological evaluation of novel FXIa inhibitors with 2-phenyl-1H-imidazole-5-carboxamide moiety as P1 fragment. Eur J Med Chem220:0 (2021) [PubMed] Article
Lei, Y; Zhang, B; Zhang, Y; Dai, X; Duan, Y; Mao, Q; Gao, J; Yang, Y; Bao, Z; Fu, X; Ping, K; Yan, C; Mou, Y; Wang, S Design, synthesis and biological evaluation of novel FXIa inhibitors with 2-phenyl-1H-imidazole-5-carboxamide moiety as P1 fragment. Eur J Med Chem220:0 (2021) [PubMed] Article