| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin G/H synthase 1 |
|---|
| Ligand | BDBM50138449 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_159406 |
|---|
| IC50 | 100000±n/a nM |
|---|
| Citation |  Singh, SK; Reddy, PG; Rao, KS; Lohray, BB; Misra, P; Rajjak, SA; Rao, YK; Venkateswarlu, A Polar substitutions in the benzenesulfonamide ring of celecoxib afford a potent 1,5-diarylpyrazole class of COX-2 inhibitors. Bioorg Med Chem Lett14:499-504 (2003) [PubMed] Singh, SK; Reddy, PG; Rao, KS; Lohray, BB; Misra, P; Rajjak, SA; Rao, YK; Venkateswarlu, A Polar substitutions in the benzenesulfonamide ring of celecoxib afford a potent 1,5-diarylpyrazole class of COX-2 inhibitors. Bioorg Med Chem Lett14:499-504 (2003) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin G/H synthase 1 |
|---|
| Name: | Prostaglandin G/H synthase 1 |
|---|
| Synonyms: | COX1 | Cyclooxygenase-1 | Cyclooxygenase-1 (COX-1) | PGH1_SHEEP | PTGS1 | Prostaglandin G/H synthase (cyclooxygenase) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 68868.60 |
|---|
| Organism: | Ovis aries (Sheep) |
|---|
| Description: | n/a |
|---|
| Residue: | 600 |
|---|
| Sequence: | MSRQSISLRFPLLLLLLSPSPVFSADPGAPAPVNPCCYYPCQHQGICVRFGLDRYQCDCT
RTGYSGPNCTIPEIWTWLRTTLRPSPSFIHFLLTHGRWLWDFVNATFIRDTLMRLVLTVR
SNLIPSPPTYNIAHDYISWESFSNVSYYTRILPSVPRDCPTPMDTKGKKQLPDAEFLSRR
FLLRRKFIPDPQSTNLMFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDNLER
QYQLRLFKDGKLKYQMLNGEVYPPSVEEAPVLMHYPRGIPPQSQMAVGQEVFGLLPGLML
YATIWLREHNRVCDLLKAEHPTWGDEQLFQTARLILIGETIKIVIEEYVQQLSGYFLQLK
FDPELLFGAQFQYRNRIAMEFNQLYHWHPLMPDSFRVGPQDYSYEQFLFNTSMLVDYGVE
ALVDAFSRQPAGRIGGGRNIDHHILHVAVDVIKESRVLRLQPFNEYRKRFGMKPYTSFQE
LTGEKEMAAELEELYGDIDALEFYPGLLLEKCHPNSIFGESMIEMGAPFSLKGLLGNPIC
SPEYWKASTFGGEVGFNLVKTATLKKLVCLNTKTCPYVSFHVPDPRQEDRPGVERPPTEL
|
|
|
|---|
| BDBM50138449 |
|---|
| n/a |
|---|
| Name | BDBM50138449 |
|---|
| Synonyms: | CHEMBL146465 | N-{5-[5-(4-Chloro-phenyl)-3-trifluoromethyl-pyrazol-1-yl]-2-sulfamoyl-benzyl}-acetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H16ClF3N4O3S |
|---|
| Mol. Mass. | 472.869 |
|---|
| SMILES | CC(=O)NCc1cc(ccc1S(N)(=O)=O)-n1nc(cc1-c1ccc(Cl)cc1)C(F)(F)F |
|---|
| Structure | 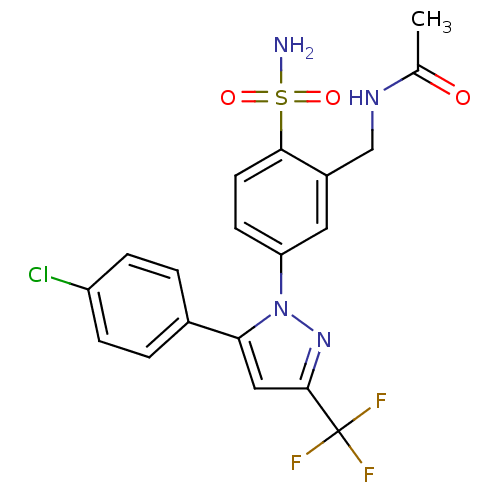
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Singh, SK; Reddy, PG; Rao, KS; Lohray, BB; Misra, P; Rajjak, SA; Rao, YK; Venkateswarlu, A Polar substitutions in the benzenesulfonamide ring of celecoxib afford a potent 1,5-diarylpyrazole class of COX-2 inhibitors. Bioorg Med Chem Lett14:499-504 (2003) [PubMed]
Singh, SK; Reddy, PG; Rao, KS; Lohray, BB; Misra, P; Rajjak, SA; Rao, YK; Venkateswarlu, A Polar substitutions in the benzenesulfonamide ring of celecoxib afford a potent 1,5-diarylpyrazole class of COX-2 inhibitors. Bioorg Med Chem Lett14:499-504 (2003) [PubMed]