| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2D6 |
|---|
| Ligand | BDBM50315305 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2135900 (CHEMBL4845510) |
|---|
| IC50 | 1640±n/a nM |
|---|
| Citation |  Nguyen, HH; Tahirovic, YA; Truax, VM; Wilson, RJ; Jecs, E; Miller, EJ; Kim, MB; Akins, NS; Xu, L; Jiang, Y; Wang, T; Sum, CS; Cvijic, ME; Schroeder, GM; Wilson, LJ; Liotta, DC Amino-Heterocycle Tetrahydroisoquinoline CXCR4 Antagonists with Improved ADME Profiles via Late-Stage Buchwald Couplings. ACS Med Chem Lett12:1605-1612 (2021) [PubMed] Article Nguyen, HH; Tahirovic, YA; Truax, VM; Wilson, RJ; Jecs, E; Miller, EJ; Kim, MB; Akins, NS; Xu, L; Jiang, Y; Wang, T; Sum, CS; Cvijic, ME; Schroeder, GM; Wilson, LJ; Liotta, DC Amino-Heterocycle Tetrahydroisoquinoline CXCR4 Antagonists with Improved ADME Profiles via Late-Stage Buchwald Couplings. ACS Med Chem Lett12:1605-1612 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2D6 |
|---|
| Name: | Cytochrome P450 2D6 |
|---|
| Synonyms: | CP2D6_HUMAN | CYP2D6 | CYP2DL1 | CYPIID6 | Cytochrome P450 2D6 (CYP2D6) | Debrisoquine 4-hydroxylase | P450-DB1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55774.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P10635 |
|---|
| Residue: | 497 |
|---|
| Sequence: | MGLEALVPLAVIVAIFLLLVDLMHRRQRWAARYPPGPLPLPGLGNLLHVDFQNTPYCFDQ
LRRRFGDVFSLQLAWTPVVVLNGLAAVREALVTHGEDTADRPPVPITQILGFGPRSQGVF
LARYGPAWREQRRFSVSTLRNLGLGKKSLEQWVTEEAACLCAAFANHSGRPFRPNGLLDK
AVSNVIASLTCGRRFEYDDPRFLRLLDLAQEGLKEESGFLREVLNAVPVLLHIPALAGKV
LRFQKAFLTQLDELLTEHRMTWDPAQPPRDLTEAFLAEMEKAKGNPESSFNDENLRIVVA
DLFSAGMVTTSTTLAWGLLLMILHPDVQRRVQQEIDDVIGQVRRPEMGDQAHMPYTTAVI
HEVQRFGDIVPLGVTHMTSRDIEVQGFRIPKGTTLITNLSSVLKDEAVWEKPFRFHPEHF
LDAQGHFVKPEAFLPFSAGRRACLGEPLARMELFLFFTSLLQHFSFSVPTGQPRPSHHGV
FAFLVSPSPYELCAVPR
|
|
|
|---|
| BDBM50315305 |
|---|
| n/a |
|---|
| Name | BDBM50315305 |
|---|
| Synonyms: | (S)-N-((1H-benzo[d]imidazol-2-yl)methyl)-N-(4-aminobutyl)-5,6,7,8-tetrahydroquinolin-8-amine | (S)-N1-((1H-benzo[d]imidazol-2-yl)methyl)-N1-(5,6,7,8-tetrahydroquinolin-8-yl)butane-1,4-diamine | CHEMBL518924 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H27N5 |
|---|
| Mol. Mass. | 349.4726 |
|---|
| SMILES | NCCCCN(Cc1nc2ccccc2[nH]1)[C@H]1CCCc2cccnc12 |r| |
|---|
| Structure | 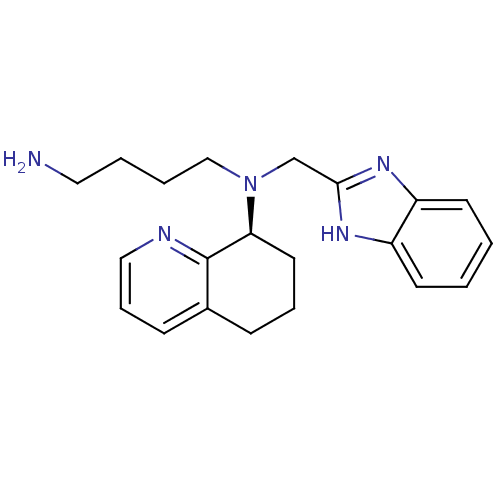
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nguyen, HH; Tahirovic, YA; Truax, VM; Wilson, RJ; Jecs, E; Miller, EJ; Kim, MB; Akins, NS; Xu, L; Jiang, Y; Wang, T; Sum, CS; Cvijic, ME; Schroeder, GM; Wilson, LJ; Liotta, DC Amino-Heterocycle Tetrahydroisoquinoline CXCR4 Antagonists with Improved ADME Profiles via Late-Stage Buchwald Couplings. ACS Med Chem Lett12:1605-1612 (2021) [PubMed] Article
Nguyen, HH; Tahirovic, YA; Truax, VM; Wilson, RJ; Jecs, E; Miller, EJ; Kim, MB; Akins, NS; Xu, L; Jiang, Y; Wang, T; Sum, CS; Cvijic, ME; Schroeder, GM; Wilson, LJ; Liotta, DC Amino-Heterocycle Tetrahydroisoquinoline CXCR4 Antagonists with Improved ADME Profiles via Late-Stage Buchwald Couplings. ACS Med Chem Lett12:1605-1612 (2021) [PubMed] Article