| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Plasminogen |
|---|
| Ligand | BDBM50157090 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_302472 (CHEMBL826337) |
|---|
| Ki | 1302±n/a nM |
|---|
| Citation |  Bruncko, M; McClellan, WJ; Wendt, MD; Sauer, DR; Geyer, A; Dalton, CR; Kaminski, MA; Weitzberg, M; Gong, J; Dellaria, JF; Mantei, R; Zhao, X; Nienaber, VL; Stewart, K; Klinghofer, V; Bouska, J; Rockway, TW; Giranda, VL Naphthamidine urokinase plasminogen activator inhibitors with improved pharmacokinetic properties. Bioorg Med Chem Lett15:93-8 (2004) [PubMed] Article Bruncko, M; McClellan, WJ; Wendt, MD; Sauer, DR; Geyer, A; Dalton, CR; Kaminski, MA; Weitzberg, M; Gong, J; Dellaria, JF; Mantei, R; Zhao, X; Nienaber, VL; Stewart, K; Klinghofer, V; Bouska, J; Rockway, TW; Giranda, VL Naphthamidine urokinase plasminogen activator inhibitors with improved pharmacokinetic properties. Bioorg Med Chem Lett15:93-8 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Plasminogen |
|---|
| Name: | Plasminogen |
|---|
| Synonyms: | Activation peptide | Angiostatin | PLG | PLMN_HUMAN | Plasmin | Plasmin heavy chain A | Plasmin heavy chain A, short form | Plasmin light chain B |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 90579.18 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 810 |
|---|
| Sequence: | MEHKEVVLLLLLFLKSGQGEPLDDYVNTQGASLFSVTKKQLGAGSIEECAAKCEEDEEFT
CRAFQYHSKEQQCVIMAENRKSSIIIRMRDVVLFEKKVYLSECKTGNGKNYRGTMSKTKN
GITCQKWSSTSPHRPRFSPATHPSEGLEENYCRNPDNDPQGPWCYTTDPEKRYDYCDILE
CEEECMHCSGENYDGKISKTMSGLECQAWDSQSPHAHGYIPSKFPNKNLKKNYCRNPDRE
LRPWCFTTDPNKRWELCDIPRCTTPPPSSGPTYQCLKGTGENYRGNVAVTVSGHTCQHWS
AQTPHTHNRTPENFPCKNLDENYCRNPDGKRAPWCHTTNSQVRWEYCKIPSCDSSPVSTE
QLAPTAPPELTPVVQDCYHGDGQSYRGTSSTTTTGKKCQSWSSMTPHRHQKTPENYPNAG
LTMNYCRNPDADKGPWCFTTDPSVRWEYCNLKKCSGTEASVVAPPPVVLLPDVETPSEED
CMFGNGKGYRGKRATTVTGTPCQDWAAQEPHRHSIFTPETNPRAGLEKNYCRNPDGDVGG
PWCYTTNPRKLYDYCDVPQCAAPSFDCGKPQVEPKKCPGRVVGGCVAHPHSWPWQVSLRT
RFGMHFCGGTLISPEWVLTAAHCLEKSPRPSSYKVILGAHQEVNLEPHVQEIEVSRLFLE
PTRKDIALLKLSSPAVITDKVIPACLPSPNYVVADRTECFITGWGETQGTFGAGLLKEAQ
LPVIENKVCNRYEFLNGRVQSTELCAGHLAGGTDSCQGDSGGPLVCFEKDKYILQGVTSW
GLGCARPNKPGVYVRVSRFVTWIEGVMRNN
|
|
|
|---|
| BDBM50157090 |
|---|
| n/a |
|---|
| Name | BDBM50157090 |
|---|
| Synonyms: | 6-(2-(1-isopropyl-2-methyl-1,2,3,4-tetrahydroisoquinolin-7-yl)cyclopropyl)-2-naphthimidamide | 6-[2-(1-Isopropyl-2-methyl-1,2,3,4-tetrahydro-isoquinolin-7-yl)-cyclopropyl]-naphthalene-2-carboxamidine | CHEMBL185515 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H31N3 |
|---|
| Mol. Mass. | 397.5551 |
|---|
| SMILES | CC(C)C1N(C)CCc2ccc(cc12)C1CC1c1ccc2cc(ccc2c1)C(N)=N |
|---|
| Structure | 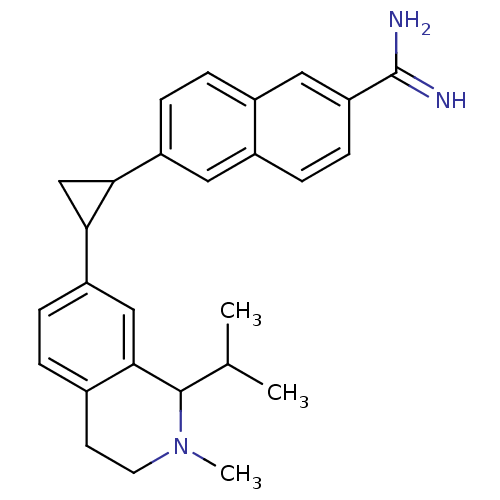
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bruncko, M; McClellan, WJ; Wendt, MD; Sauer, DR; Geyer, A; Dalton, CR; Kaminski, MA; Weitzberg, M; Gong, J; Dellaria, JF; Mantei, R; Zhao, X; Nienaber, VL; Stewart, K; Klinghofer, V; Bouska, J; Rockway, TW; Giranda, VL Naphthamidine urokinase plasminogen activator inhibitors with improved pharmacokinetic properties. Bioorg Med Chem Lett15:93-8 (2004) [PubMed] Article
Bruncko, M; McClellan, WJ; Wendt, MD; Sauer, DR; Geyer, A; Dalton, CR; Kaminski, MA; Weitzberg, M; Gong, J; Dellaria, JF; Mantei, R; Zhao, X; Nienaber, VL; Stewart, K; Klinghofer, V; Bouska, J; Rockway, TW; Giranda, VL Naphthamidine urokinase plasminogen activator inhibitors with improved pharmacokinetic properties. Bioorg Med Chem Lett15:93-8 (2004) [PubMed] Article