| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Bifunctional purine biosynthesis protein ATIC |
|---|
| Ligand | BDBM50158378 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_430370 (CHEMBL914875) |
|---|
| IC50 | 600±n/a nM |
|---|
| Citation |  Li, C; Xu, L; Wolan, DW; Wilson, IA; Olson, AJ Virtual screening of human 5-aminoimidazole-4-carboxamide ribonucleotide transformylase against the NCI diversity set by use of AutoDock to identify novel nonfolate inhibitors. J Med Chem47:6681-90 (2004) [PubMed] Article Li, C; Xu, L; Wolan, DW; Wilson, IA; Olson, AJ Virtual screening of human 5-aminoimidazole-4-carboxamide ribonucleotide transformylase against the NCI diversity set by use of AutoDock to identify novel nonfolate inhibitors. J Med Chem47:6681-90 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Bifunctional purine biosynthesis protein ATIC |
|---|
| Name: | Bifunctional purine biosynthesis protein ATIC |
|---|
| Synonyms: | 5-aminoimidazole-4-carboxamide ribonucleotide formyltransferase | 5-aminoimidazole-4-carboxamide-ribonucleotide transformylase | AICAR Tfase | AICAR transformylase | ATIC | Aminoimidazole carboxamide ribonucleotide transformylase (AICAR Tfase) | Bifunctional purine biosynthesis protein PURH | IMP Cyclohydrolase (IMPCH) | IMP cyclohydrolase | IMP synthetase | Inosinicase | PUR9_HUMAN | PURH | Phosphoribosylaminoimidazolecarboxamide formyltransferase | Thymidylate synthase/GAR transformylase/AICAR transformylase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 64616.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P31939 |
|---|
| Residue: | 592 |
|---|
| Sequence: | MAPGQLALFSVSDKTGLVEFARNLTALGLNLVASGGTAKALRDAGLAVRDVSELTGFPEM
LGGRVKTLHPAVHAGILARNIPEDNADMARLDFNLIRVVACNLYPFVKTVASPGVTVEEA
VEQIDIGGVTLLRAAAKNHARVTVVCEPEDYVVVSTEMQSSESKDTSLETRRQLALKAFT
HTAQYDEAISDYFRKQYSKGVSQMPLRYGMNPHQTPAQLYTLQPKLPITVLNGAPGFINL
CDALNAWQLVKELKEALGIPAAASFKHVSPAGAAVGIPLSEDEAKVCMVYDLYKTLTPIS
AAYARARGADRMSSFGDFVALSDVCDVPTAKIISREVSDGIIAPGYEEEALTILSKKKNG
NYCVLQMDQSYKPDENEVRTLFGLHLSQKRNNGVVDKSLFSNVVTKNKDLPESALRDLIV
ATIAVKYTQSNSVCYAKNGQVIGIGAGQQSRIHCTRLAGDKANYWWLRHHPQVLSMKFKT
GVKRAEISNAIDQYVTGTIGEDEDLIKWKALFEEVPELLTEAEKKEWVEKLTEVSISSDA
FFPFRDNVDRAKRSGVAYIAAPSGSAADKVVIEACDELGIILAHTNLRLFHH
|
|
|
|---|
| BDBM50158378 |
|---|
| n/a |
|---|
| Name | BDBM50158378 |
|---|
| Synonyms: | 5-(5-sulfamoyl-naphthalen-2-ylazo)-naphthalene-1-sulfonic acid | CHEMBL375938 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H15N3O5S2 |
|---|
| Mol. Mass. | 441.48 |
|---|
| SMILES | NS(=O)(=O)c1cccc2cc(ccc12)\N=N\c1cccc2c(cccc12)S(O)(=O)=O |
|---|
| Structure | 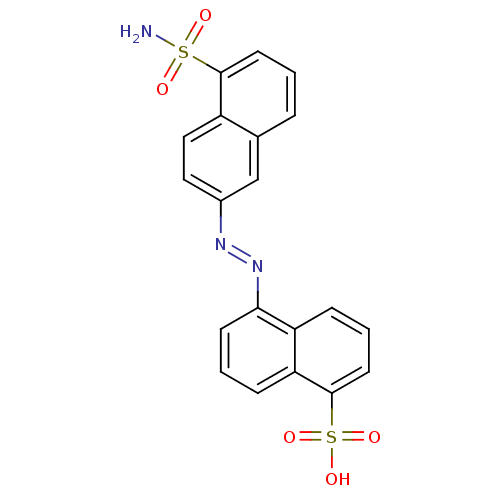
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Li, C; Xu, L; Wolan, DW; Wilson, IA; Olson, AJ Virtual screening of human 5-aminoimidazole-4-carboxamide ribonucleotide transformylase against the NCI diversity set by use of AutoDock to identify novel nonfolate inhibitors. J Med Chem47:6681-90 (2004) [PubMed] Article
Li, C; Xu, L; Wolan, DW; Wilson, IA; Olson, AJ Virtual screening of human 5-aminoimidazole-4-carboxamide ribonucleotide transformylase against the NCI diversity set by use of AutoDock to identify novel nonfolate inhibitors. J Med Chem47:6681-90 (2004) [PubMed] Article