| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Melanocortin receptor 4 |
|---|
| Ligand | BDBM50134486 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_430483 (CHEMBL917732) |
|---|
| EC50 | 110±n/a nM |
|---|
| Citation |  Chen, C; Pontillo, J; Fleck, BA; Gao, Y; Wen, J; Tran, JA; Tucci, FC; Marinkovic, D; Foster, AC; Saunders, J 4-{(2R)-[3-Aminopropionylamido]-3-(2,4-dichlorophenyl)propionyl}-1-{2-[(2-thienyl)ethylaminomethyl]phenyl}piperazine as a potent and selective melanocortin-4 receptor antagonist--design, synthesis, and characterization. J Med Chem47:6821-30 (2004) [PubMed] Article Chen, C; Pontillo, J; Fleck, BA; Gao, Y; Wen, J; Tran, JA; Tucci, FC; Marinkovic, D; Foster, AC; Saunders, J 4-{(2R)-[3-Aminopropionylamido]-3-(2,4-dichlorophenyl)propionyl}-1-{2-[(2-thienyl)ethylaminomethyl]phenyl}piperazine as a potent and selective melanocortin-4 receptor antagonist--design, synthesis, and characterization. J Med Chem47:6821-30 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Melanocortin receptor 4 |
|---|
| Name: | Melanocortin receptor 4 |
|---|
| Synonyms: | MC4-R | MC4R | MC4R_HUMAN | Melanocortin MC4 | Melanocortin receptor 4 (MC-4) | Melanocortin receptor 4 (MC4-R) | Melanocortin receptor 4 (MC4R) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 36949.50 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P32245 |
|---|
| Residue: | 332 |
|---|
| Sequence: | MVNSTHRGMHTSLHLWNRSSYRLHSNASESLGKGYSDGGCYEQLFVSPEVFVTLGVISLL
ENILVIVAIAKNKNLHSPMYFFICSLAVADMLVSVSNGSETIVITLLNSTDTDAQSFTVN
IDNVIDSVICSSLLASICSLLSIAVDRYFTIFYALQYHNIMTVKRVGIIISCIWAACTVS
GILFIIYSDSSAVIICLITMFFTMLALMASLYVHMFLMARLHIKRIAVLPGTGAIRQGAN
MKGAITLTILIGVFVVCWAPFFLHLIFYISCPQNPYCVCFMSHFNLYLILIMCNSIIDPL
IYALRSQELRKTFKEIICCYPLGGLCDLSSRY
|
|
|
|---|
| BDBM50134486 |
|---|
| n/a |
|---|
| Name | BDBM50134486 |
|---|
| Synonyms: | (R)-1,2,3,4-Tetrahydro-isoquinoline-3-carboxylic acid {(R)-1-(4-chloro-benzyl)-2-oxo-2-[4-(2-[1,2,4]triazol-1-ylmethyl-phenyl)-piperazin-1-yl]-ethyl}-amide | (R)-N-((R)-1-(4-(2-((1H-1,2,4-triazol-1-yl)methyl)phenyl)piperazin-1-yl)-3-(4-chlorophenyl)-1-oxopropan-2-yl)-1,2,3,4-tetrahydroisoquinoline-3-carboxamide | 1,2,3,4-Tetrahydro-isoquinoline-3-carboxylic acid {(R)-1-(4-chloro-benzyl)-2-oxo-2-[4-(2-[1,2,4]triazol-1-ylmethyl-phenyl)-piperazin-1-yl]-ethyl}-amide | CHEMBL340355 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H34ClN7O2 |
|---|
| Mol. Mass. | 584.111 |
|---|
| SMILES | Clc1ccc(C[C@@H](NC(=O)[C@H]2Cc3ccccc3CN2)C(=O)N2CCN(CC2)c2ccccc2Cn2cncn2)cc1 |r| |
|---|
| Structure | 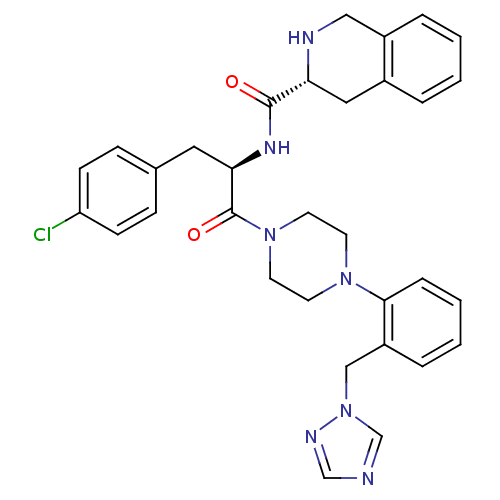
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Chen, C; Pontillo, J; Fleck, BA; Gao, Y; Wen, J; Tran, JA; Tucci, FC; Marinkovic, D; Foster, AC; Saunders, J 4-{(2R)-[3-Aminopropionylamido]-3-(2,4-dichlorophenyl)propionyl}-1-{2-[(2-thienyl)ethylaminomethyl]phenyl}piperazine as a potent and selective melanocortin-4 receptor antagonist--design, synthesis, and characterization. J Med Chem47:6821-30 (2004) [PubMed] Article
Chen, C; Pontillo, J; Fleck, BA; Gao, Y; Wen, J; Tran, JA; Tucci, FC; Marinkovic, D; Foster, AC; Saunders, J 4-{(2R)-[3-Aminopropionylamido]-3-(2,4-dichlorophenyl)propionyl}-1-{2-[(2-thienyl)ethylaminomethyl]phenyl}piperazine as a potent and selective melanocortin-4 receptor antagonist--design, synthesis, and characterization. J Med Chem47:6821-30 (2004) [PubMed] Article