| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2E1 |
|---|
| Ligand | BDBM406663 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2193432 (CHEMBL5105792) |
|---|
| IC50 | >30000±n/a nM |
|---|
| Citation |  Leit, S; Greenwood, JR; Mondal, S; Carriero, S; Dahlgren, M; Harriman, GC; Kennedy-Smith, JJ; Kapeller, R; Lawson, JP; Romero, DL; Toms, AV; Shelley, M; Wester, RT; Westlin, W; McElwee, JJ; Miao, W; Edmondson, SD; Masse, CE Potent and selective TYK2-JH1 inhibitors highly efficacious in rodent model of psoriasis. Bioorg Med Chem Lett73:0 (2022) [PubMed] Article Leit, S; Greenwood, JR; Mondal, S; Carriero, S; Dahlgren, M; Harriman, GC; Kennedy-Smith, JJ; Kapeller, R; Lawson, JP; Romero, DL; Toms, AV; Shelley, M; Wester, RT; Westlin, W; McElwee, JJ; Miao, W; Edmondson, SD; Masse, CE Potent and selective TYK2-JH1 inhibitors highly efficacious in rodent model of psoriasis. Bioorg Med Chem Lett73:0 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2E1 |
|---|
| Name: | Cytochrome P450 2E1 |
|---|
| Synonyms: | CP2E1_HUMAN | CYP2E | CYP2E1 | Cytochrome P450 2E1 (CYP2E1) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 56860.34 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05181 |
|---|
| Residue: | 493 |
|---|
| Sequence: | MSALGVTVALLVWAAFLLLVSMWRQVHSSWNLPPGPFPLPIIGNLFQLELKNIPKSFTRL
AQRFGPVFTLYVGSQRMVVMHGYKAVKEALLDYKDEFSGRGDLPAFHAHRDRGIIFNNGP
TWKDIRRFSLTTLRNYGMGKQGNESRIQREAHFLLEALRKTQGQPFDPTFLIGCAPCNVI
ADILFRKHFDYNDEKFLRLMYLFNENFHLLSTPWLQLYNNFPSFLHYLPGSHRKVIKNVA
EVKEYVSERVKEHHQSLDPNCPRDLTDCLLVEMEKEKHSAERLYTMDGITVTVADLFFAG
TETTSTTLRYGLLILMKYPEIEEKLHEEIDRVIGPSRIPAIKDRQEMPYMDAVVHEIQRF
ITLVPSNLPHEATRDTIFRGYLIPKGTVVVPTLDSVLYDNQEFPDPEKFKPEHFLNENGK
FKYSDYFKPFSTGKRVCAGEGLARMELFLLLCAILQHFNLKPLVDPKDIDLSPIHIGFGC
IPPRYKLCVIPRS
|
|
|
|---|
| BDBM406663 |
|---|
| n/a |
|---|
| Name | BDBM406663 |
|---|
| Synonyms: | US10336752, Compound 1 | US10336752, Compound 2 | US10336752, Compound 4 | US10336752, Compound 5 | US10336752, Compound 6 | US11040967, Compound 6 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H20F2N4O2 |
|---|
| Mol. Mass. | 434.438 |
|---|
| SMILES | Fc1cccc(F)c1-c1cc(Nc2ccc(cc2)C(=O)N2CCCC2)c2C(=O)NCc2n1 |
|---|
| Structure | 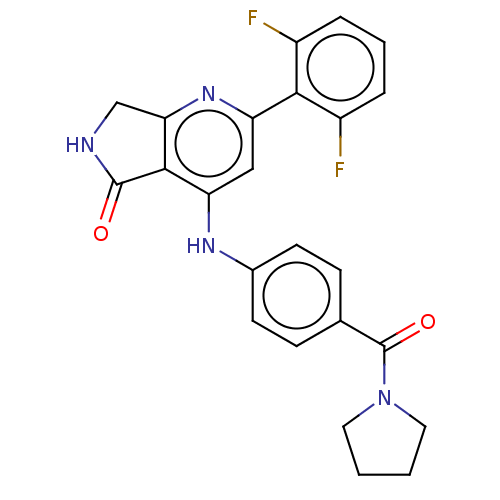
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Leit, S; Greenwood, JR; Mondal, S; Carriero, S; Dahlgren, M; Harriman, GC; Kennedy-Smith, JJ; Kapeller, R; Lawson, JP; Romero, DL; Toms, AV; Shelley, M; Wester, RT; Westlin, W; McElwee, JJ; Miao, W; Edmondson, SD; Masse, CE Potent and selective TYK2-JH1 inhibitors highly efficacious in rodent model of psoriasis. Bioorg Med Chem Lett73:0 (2022) [PubMed] Article
Leit, S; Greenwood, JR; Mondal, S; Carriero, S; Dahlgren, M; Harriman, GC; Kennedy-Smith, JJ; Kapeller, R; Lawson, JP; Romero, DL; Toms, AV; Shelley, M; Wester, RT; Westlin, W; McElwee, JJ; Miao, W; Edmondson, SD; Masse, CE Potent and selective TYK2-JH1 inhibitors highly efficacious in rodent model of psoriasis. Bioorg Med Chem Lett73:0 (2022) [PubMed] Article