

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Growth hormone secretagogue receptor type 1 | ||
| Ligand | BDBM50161058 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_306206 (CHEMBL830164) | ||
| IC50 | 350±n/a nM | ||
| Citation |  Xin, Z; Zhao, H; Serby, MD; Liu, B; Schaefer, VG; Falls, DH; Kaszubska, W; Colins, CA; Sham, HL; Liu, G Synthesis and structure-activity relationships of isoxazole carboxamides as growth hormone secretagogue receptor antagonists. Bioorg Med Chem Lett15:1201-4 (2005) [PubMed] Article Xin, Z; Zhao, H; Serby, MD; Liu, B; Schaefer, VG; Falls, DH; Kaszubska, W; Colins, CA; Sham, HL; Liu, G Synthesis and structure-activity relationships of isoxazole carboxamides as growth hormone secretagogue receptor antagonists. Bioorg Med Chem Lett15:1201-4 (2005) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Growth hormone secretagogue receptor type 1 | |||
| Name: | Growth hormone secretagogue receptor type 1 | ||
| Synonyms: | GH-releasing peptide receptor | GHRP | GHS-R | GHSR | GHSR_HUMAN | Ghrelin Receptor (Growth Hormone Secretagogue Receptor Type 1) | Ghrelin receptor | Ghrelin receptor 1a (GHS-R1a) | ||
| Type: | Receptor | ||
| Mol. Mass.: | 41334.57 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | Receptor binding studies use plasma membranes from LLC PK-1 cells transiently transfected with hGHSR1a. | ||
| Residue: | 366 | ||
| Sequence: |
| ||
| BDBM50161058 | |||
| n/a | |||
| Name | BDBM50161058 | ||
| Synonyms: | 3-(2,6-Dichloro-phenyl)-5-methyl-isoxazole-4-carboxylic acid {2-[(benzo[1,3]dioxol-5-ylmethyl)-carbamoyl]-phenyl}-amide | CHEMBL181584 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C26H19Cl2N3O5 | ||
| Mol. Mass. | 524.352 | ||
| SMILES | Cc1onc(c1C(=O)Nc1ccccc1C(=O)NCc1ccc2OCOc2c1)-c1c(Cl)cccc1Cl |(-3.38,2.35,;-4.33,1.12,;-5.86,1.2,;-6.42,-.22,;-5.22,-1.19,;-3.93,-.36,;-2.46,-.75,;-2.07,-2.24,;-1.39,.34,;.1,-.03,;.89,-1.35,;2.45,-1.34,;3.2,,;2.41,1.32,;.87,1.3,;.85,2.84,;-.23,3.93,;1.93,3.93,;3.43,4.35,;4.52,3.25,;4.09,1.79,;5.2,.7,;6.7,1.12,;7.99,.27,;9.18,1.26,;8.62,2.7,;7.08,2.59,;5.99,3.68,;-5.13,-2.73,;-3.77,-3.43,;-2.28,-3.83,;-3.67,-4.99,;-4.96,-5.81,;-6.34,-5.1,;-6.42,-3.57,;-7.8,-2.9,)| | ||
| Structure | 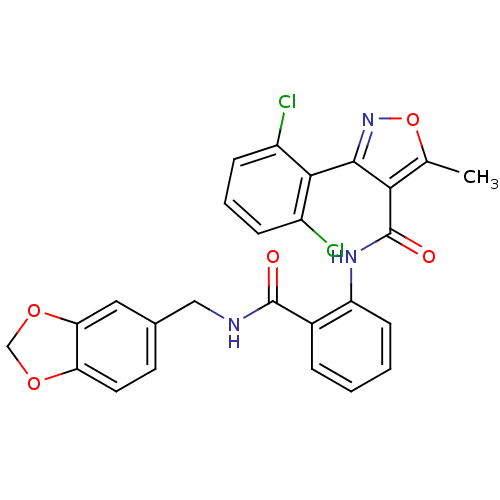
| ||