| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Liver carboxylesterase 1 |
|---|
| Ligand | BDBM50100865 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_305509 (CHEMBL831110) |
|---|
| IC50 | 1000±n/a nM |
|---|
| Citation |  Boger, DL; Miyauchi, H; Du, W; Hardouin, C; Fecik, RA; Cheng, H; Hwang, I; Hedrick, MP; Leung, D; Acevedo, O; Guimarães, CR; Jorgensen, WL; Cravatt, BF Discovery of a potent, selective, and efficacious class of reversible alpha-ketoheterocycle inhibitors of fatty acid amide hydrolase effective as analgesics. J Med Chem48:1849-56 (2005) [PubMed] Article Boger, DL; Miyauchi, H; Du, W; Hardouin, C; Fecik, RA; Cheng, H; Hwang, I; Hedrick, MP; Leung, D; Acevedo, O; Guimarães, CR; Jorgensen, WL; Cravatt, BF Discovery of a potent, selective, and efficacious class of reversible alpha-ketoheterocycle inhibitors of fatty acid amide hydrolase effective as analgesics. J Med Chem48:1849-56 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Liver carboxylesterase 1 |
|---|
| Name: | Liver carboxylesterase 1 |
|---|
| Synonyms: | Acyl coenzyme A:cholesterol acyltransferase | Acyl-CoA: cholesterol acyltransferase (ACAT) | CES1 | CES2 | EST1_HUMAN | SES1 | Serine hydrolase (CES1) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 62520.83 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 567 |
|---|
| Sequence: | MWLRAFILATLSASAAWGHPSSPPVVDTVHGKVLGKFVSLEGFAQPVAIFLGIPFAKPPL
GPLRFTPPQPAEPWSFVKNATSYPPMCTQDPKAGQLLSELFTNRKENIPLKLSEDCLYLN
IYTPADLTKKNRLPVMVWIHGGGLMVGAASTYDGLALAAHENVVVVTIQYRLGIWGFFST
GDEHSRGNWGHLDQVAALRWVQDNIASFGGNPGSVTIFGESAGGESVSVLVLSPLAKNLF
HRAISESGVALTSVLVKKGDVKPLAEQIAITAGCKTTTSAVMVHCLRQKTEEELLETTLK
MKFLSLDLQGDPRESQPLLGTVIDGMLLLKTPEELQAERNFHTVPYMVGINKQEFGWLIP
MQLMSYPLSEGQLDQKTAMSLLWKSYPLVCIAKELIPEATEKYLGGTDDTVKKKDLFLDL
IADVMFGVPSVIVARNHRDAGAPTYMYEFQYRPSFSSDMKPKTVIGDHGDELFSVFGAPF
LKEGASEEEIRLSKMVMKFWANFARNGNPNGEGLPHWPEYNQKEGYLQIGANTQAAQKLK
DKEVAFWTNLFAKKAVEKPPQTEHIEL
|
|
|
|---|
| BDBM50100865 |
|---|
| n/a |
|---|
| Name | BDBM50100865 |
|---|
| Synonyms: | (Z)-1-(oxazolo[4,5-b]pyridin-2-yl)octadec-9-en-1-one | (Z)-1-Oxazolo[4,5-b]pyridin-2-yl-octadec-9-en-1-one | 1-Oxazolo[4,5-b]pyridin-2-yl-nonadec-10-en-2-one | CHEMBL35925 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H36N2O2 |
|---|
| Mol. Mass. | 384.5548 |
|---|
| SMILES | CCCCCCCC\C=C/CCCCCCCC(=O)c1nc2ncccc2o1 |
|---|
| Structure | 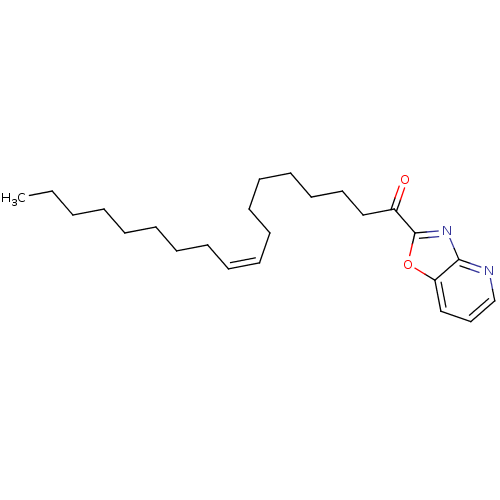
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Boger, DL; Miyauchi, H; Du, W; Hardouin, C; Fecik, RA; Cheng, H; Hwang, I; Hedrick, MP; Leung, D; Acevedo, O; Guimarães, CR; Jorgensen, WL; Cravatt, BF Discovery of a potent, selective, and efficacious class of reversible alpha-ketoheterocycle inhibitors of fatty acid amide hydrolase effective as analgesics. J Med Chem48:1849-56 (2005) [PubMed] Article
Boger, DL; Miyauchi, H; Du, W; Hardouin, C; Fecik, RA; Cheng, H; Hwang, I; Hedrick, MP; Leung, D; Acevedo, O; Guimarães, CR; Jorgensen, WL; Cravatt, BF Discovery of a potent, selective, and efficacious class of reversible alpha-ketoheterocycle inhibitors of fatty acid amide hydrolase effective as analgesics. J Med Chem48:1849-56 (2005) [PubMed] Article